General Information
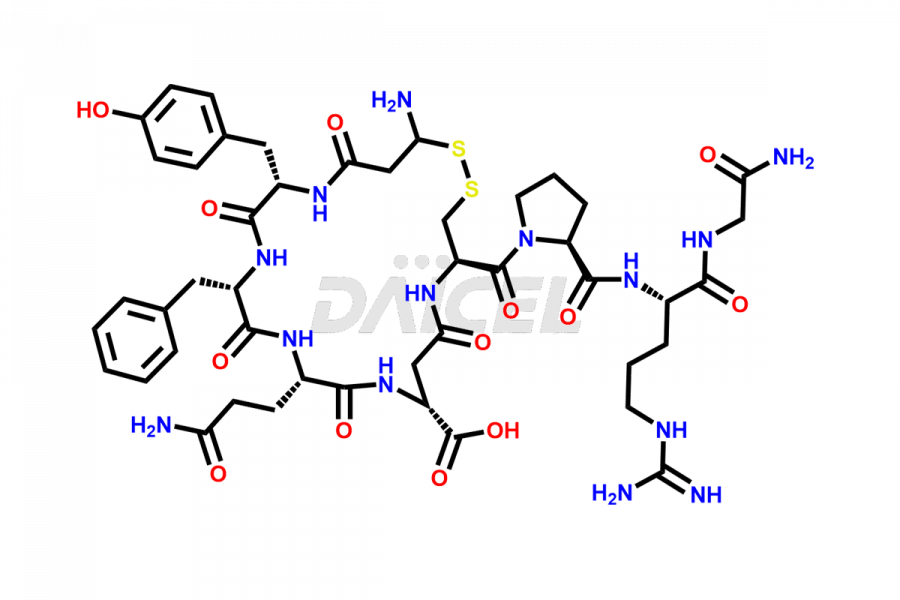
Vasopressin Impurities and Vasopressin
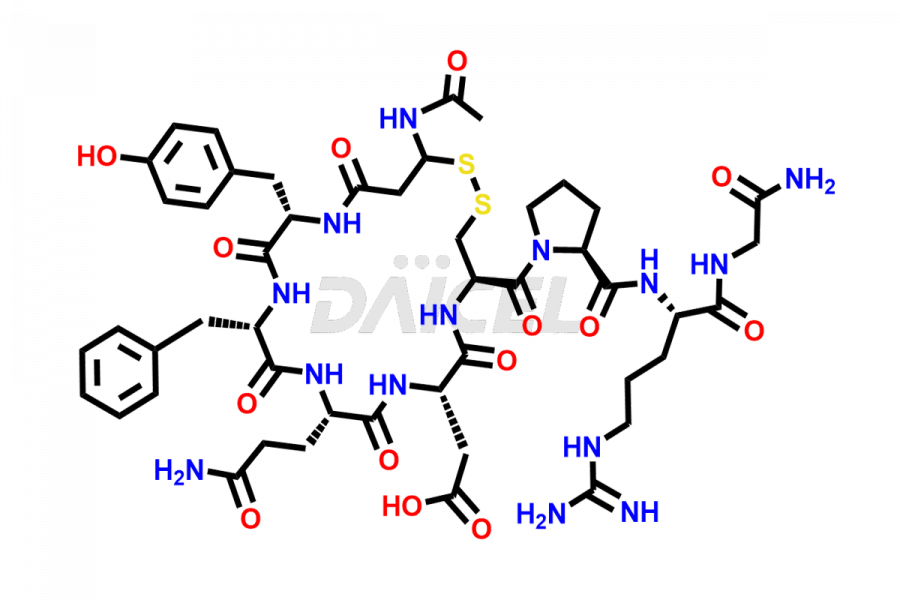
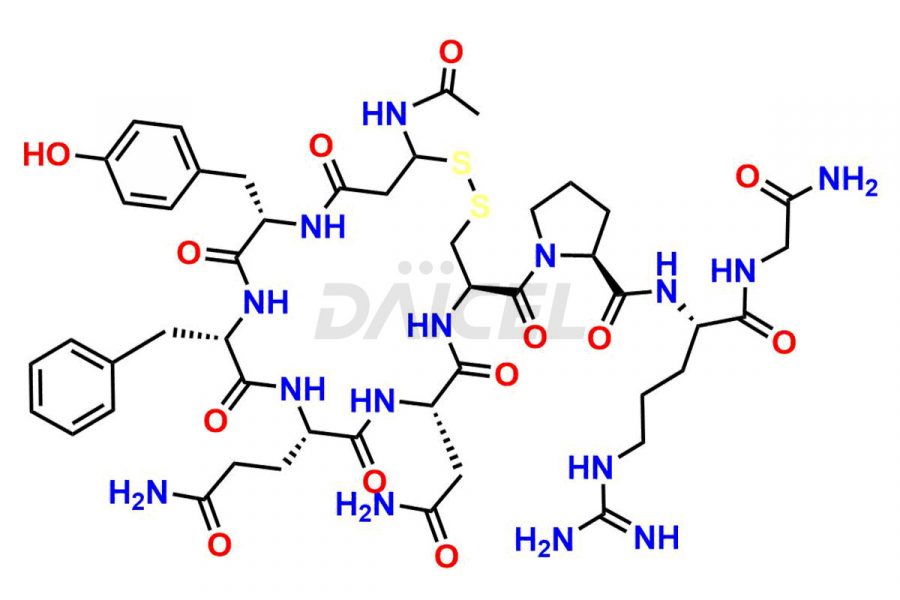
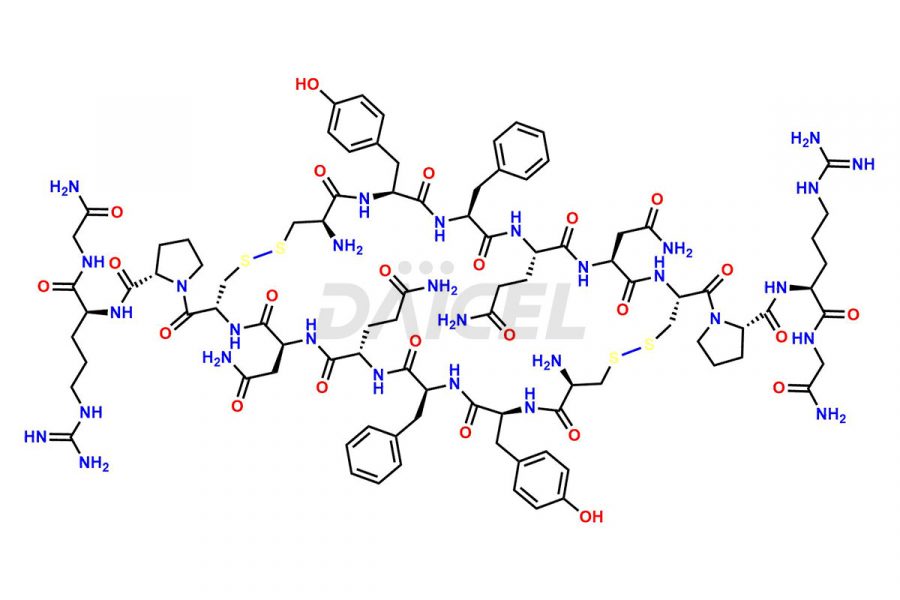
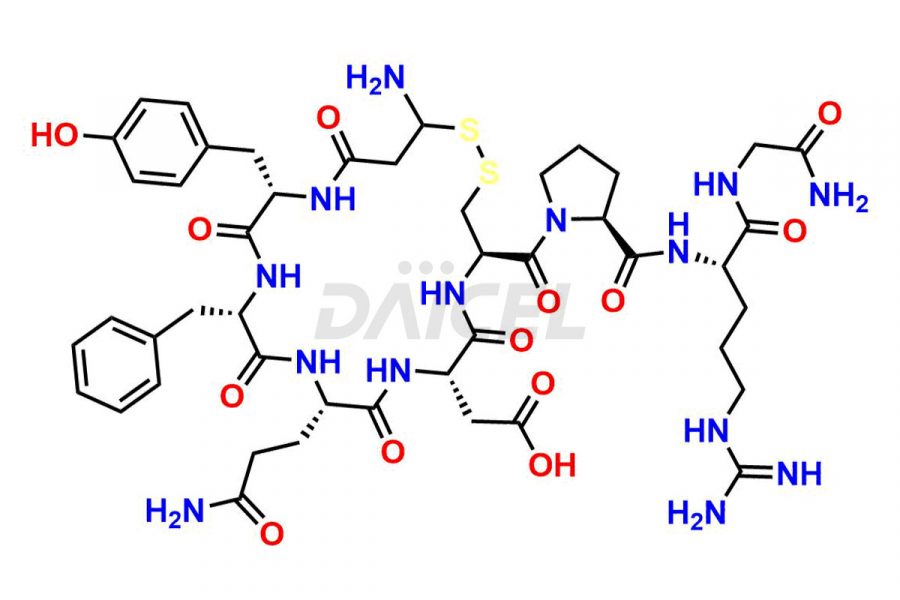
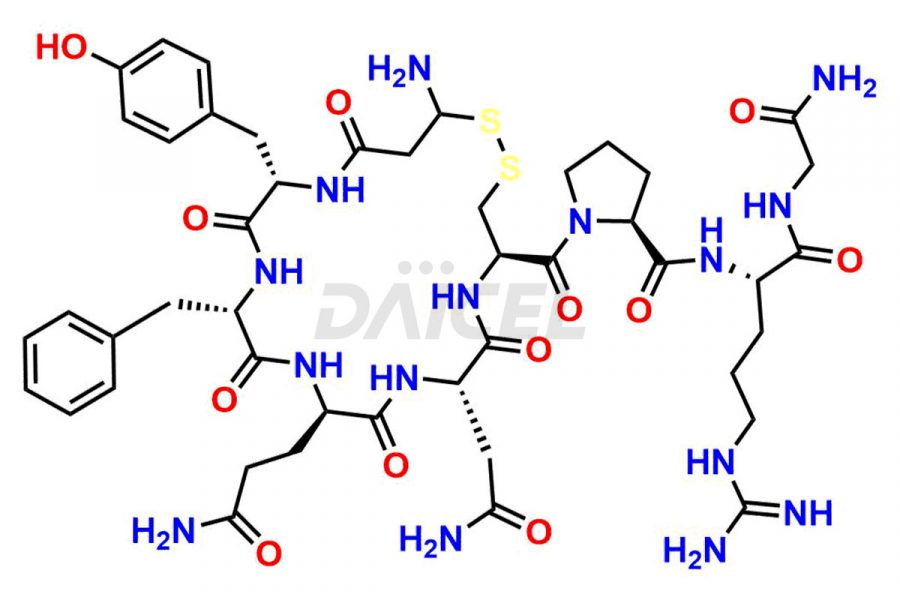
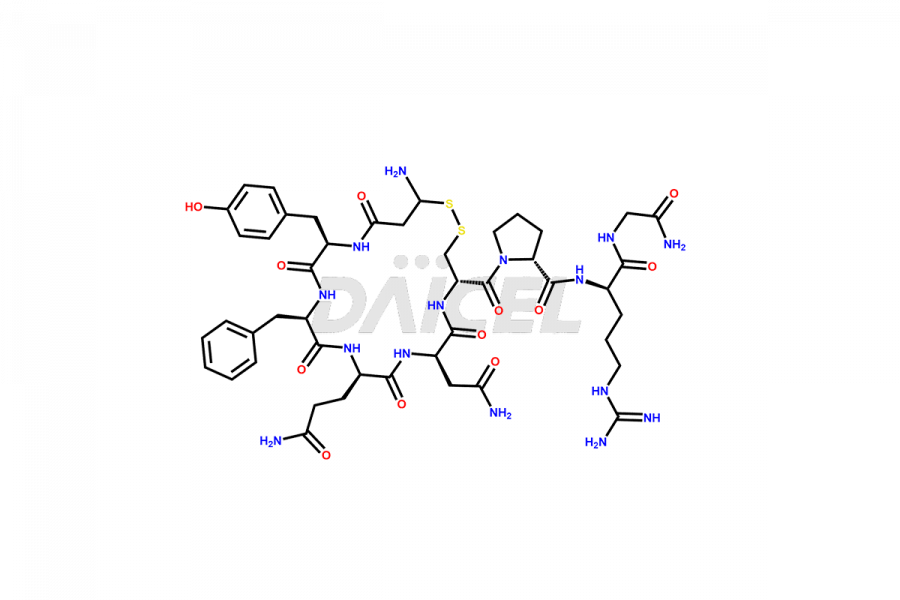
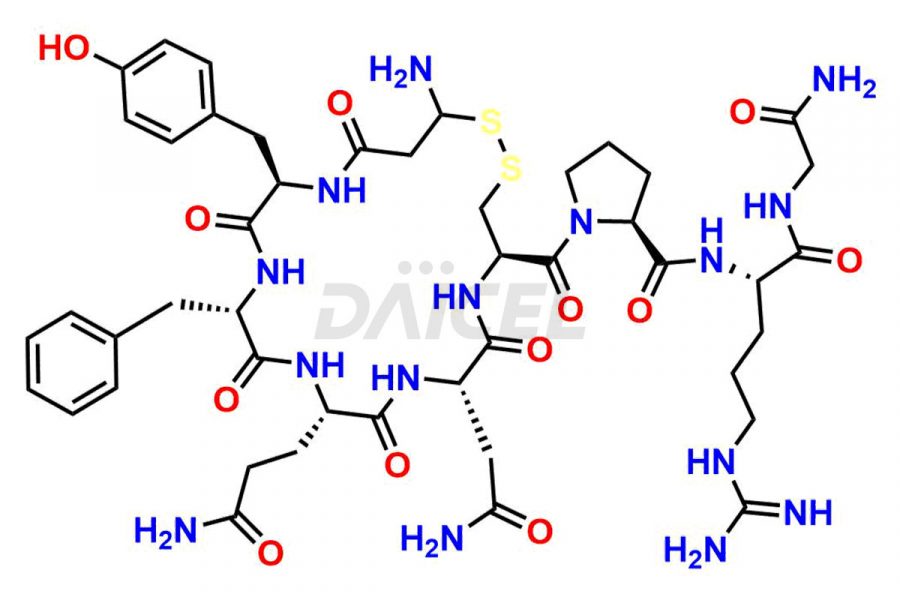
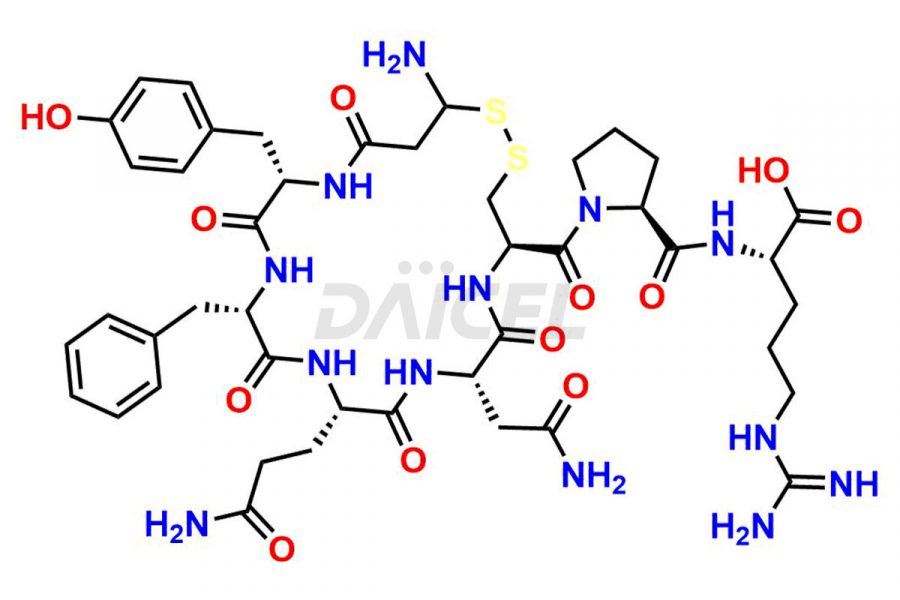
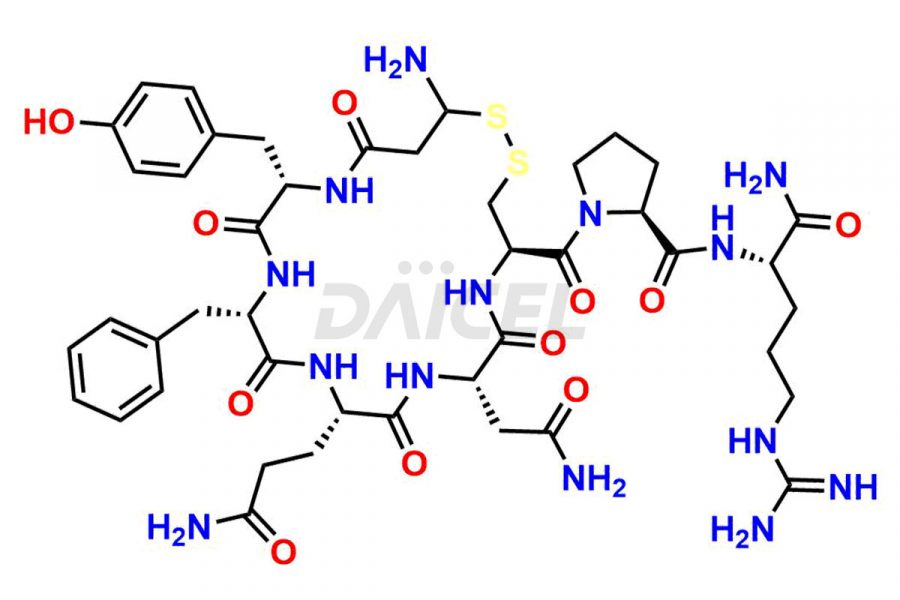
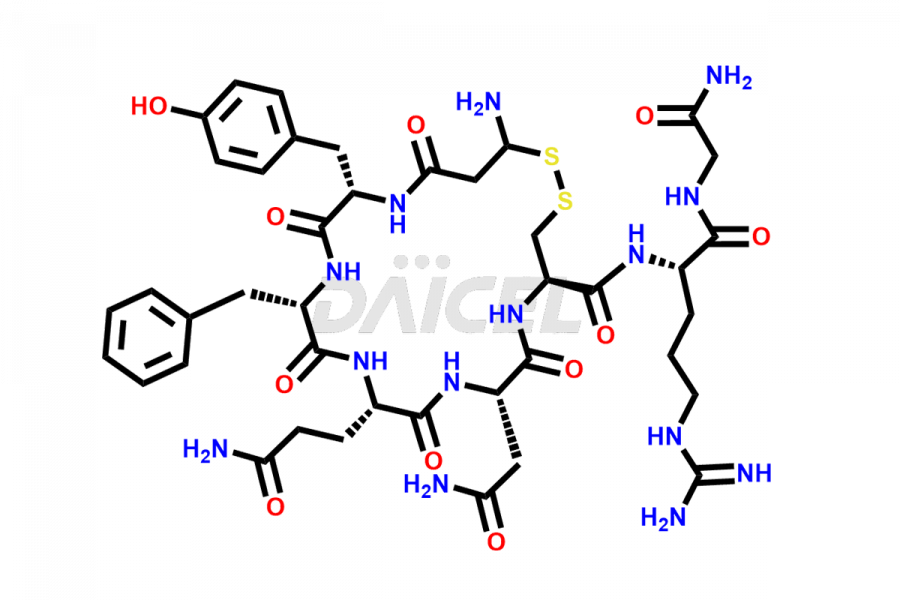
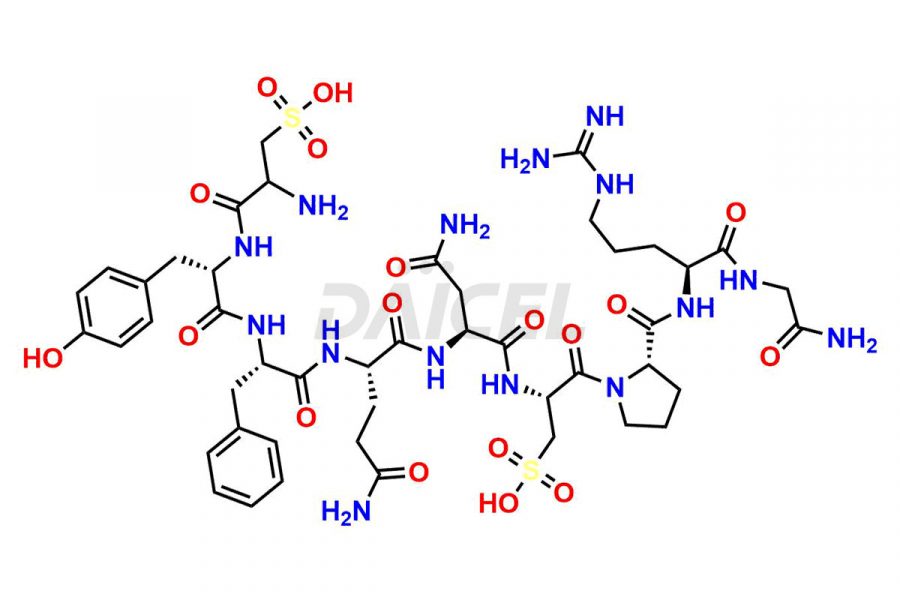
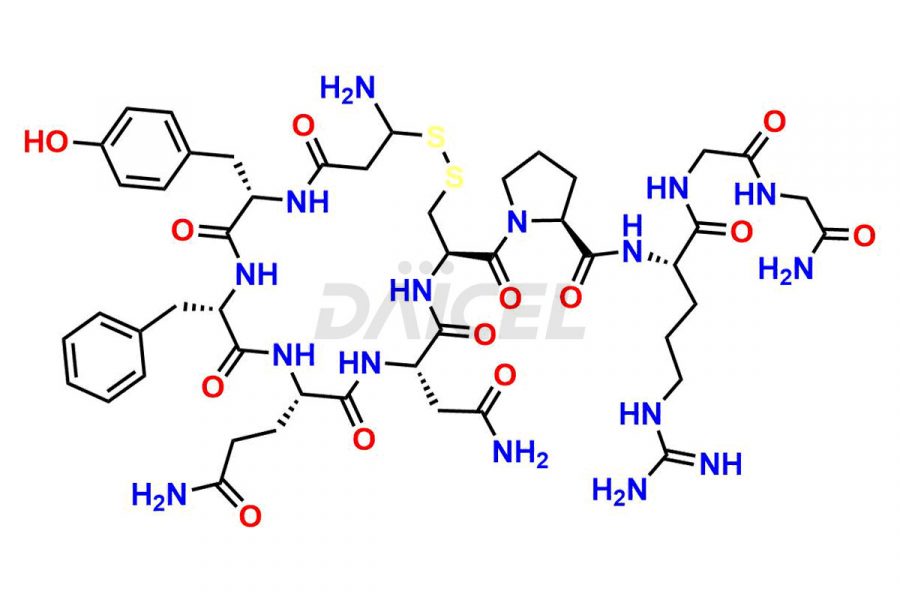
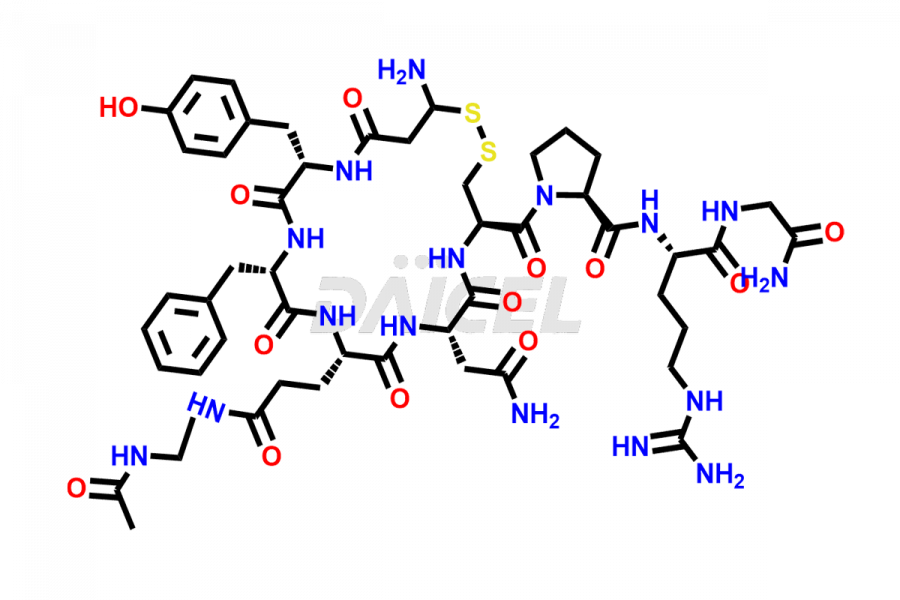
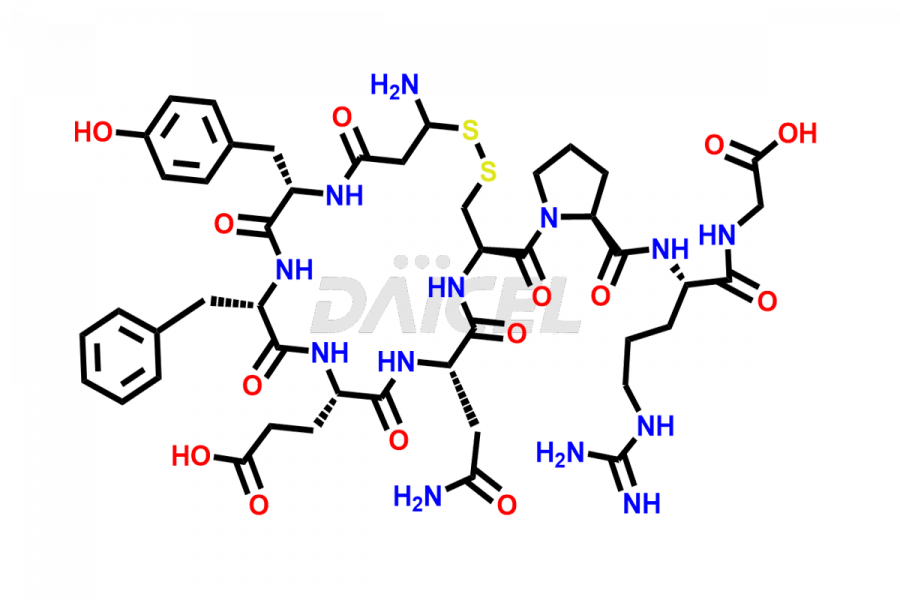
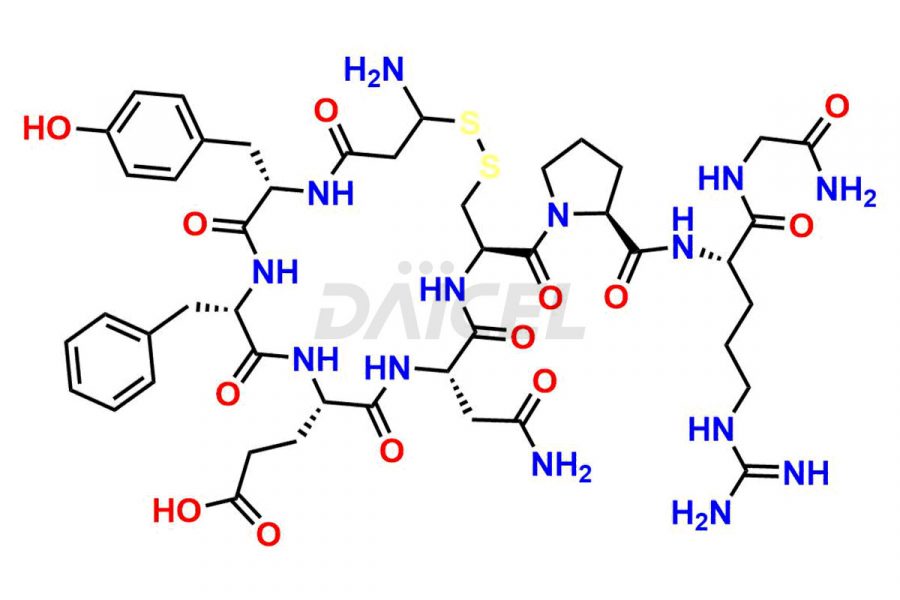
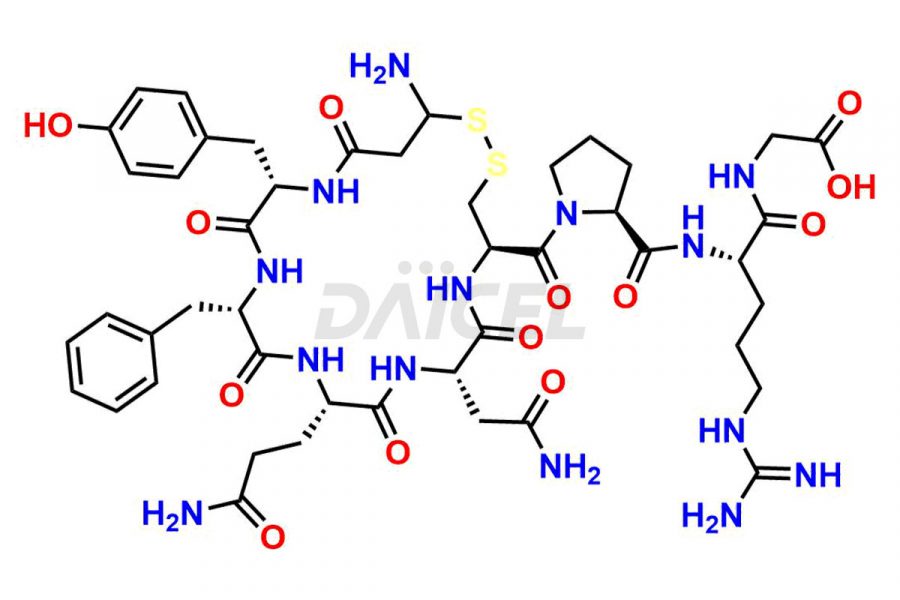
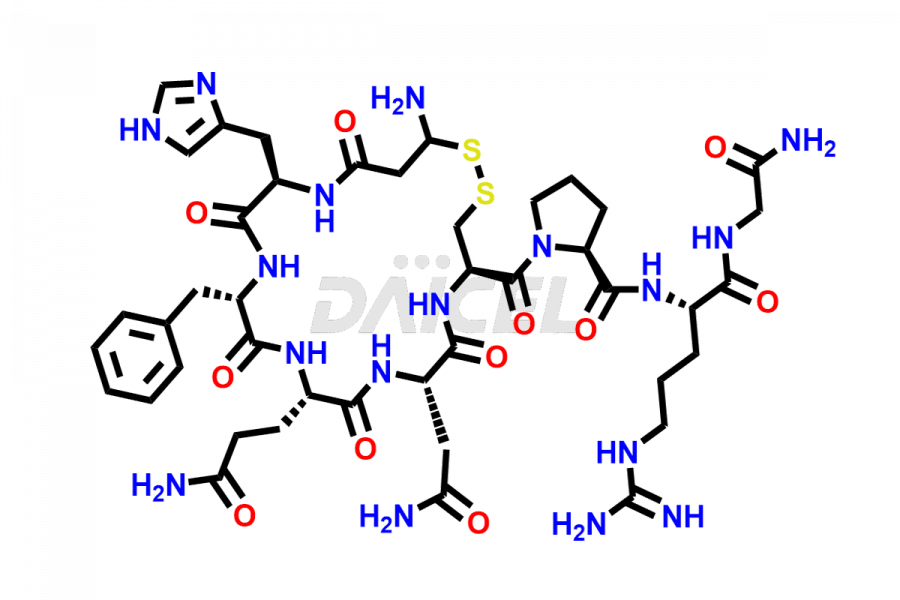
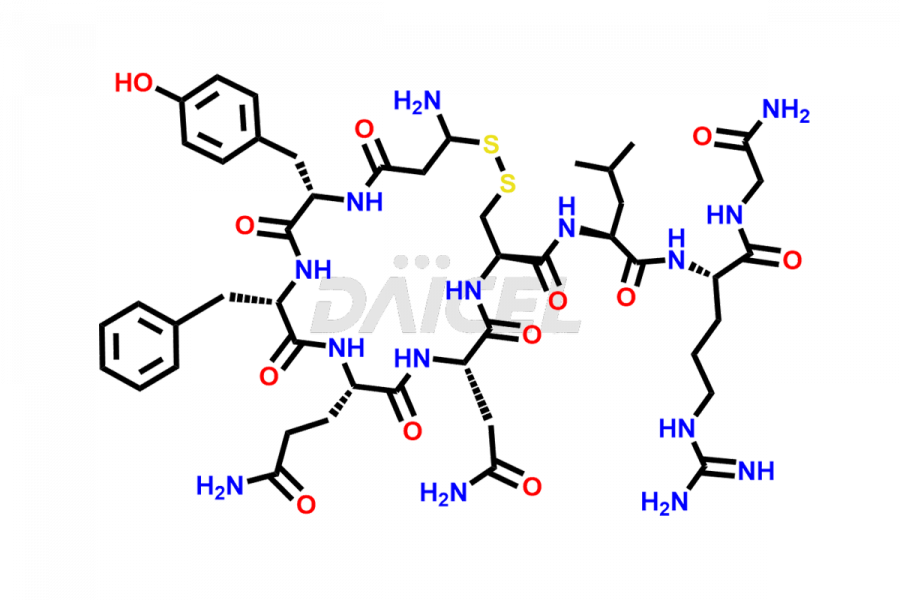
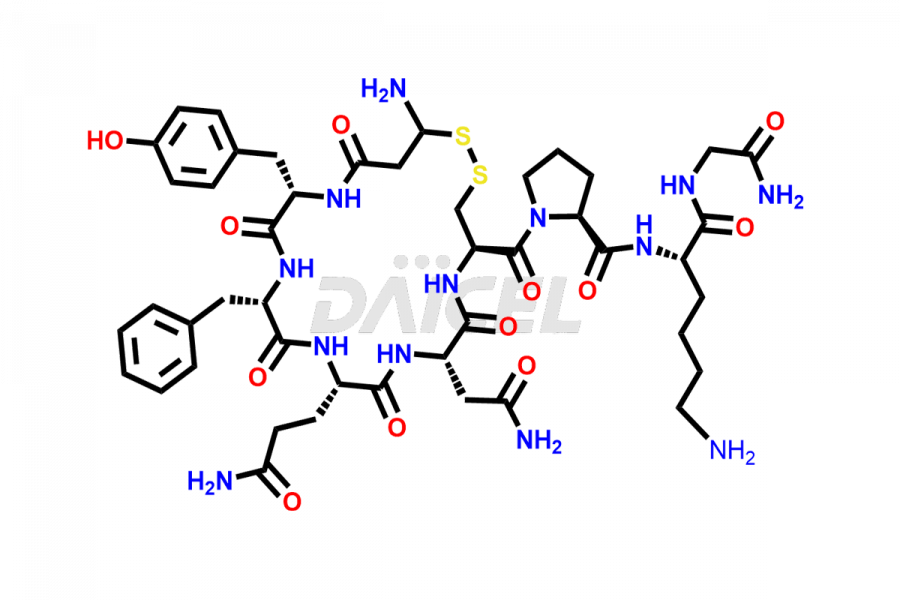
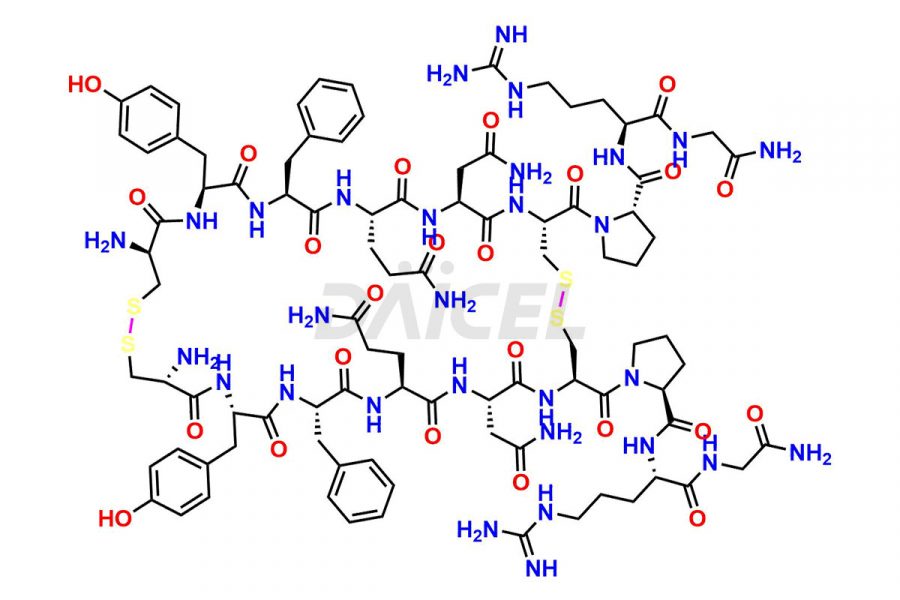
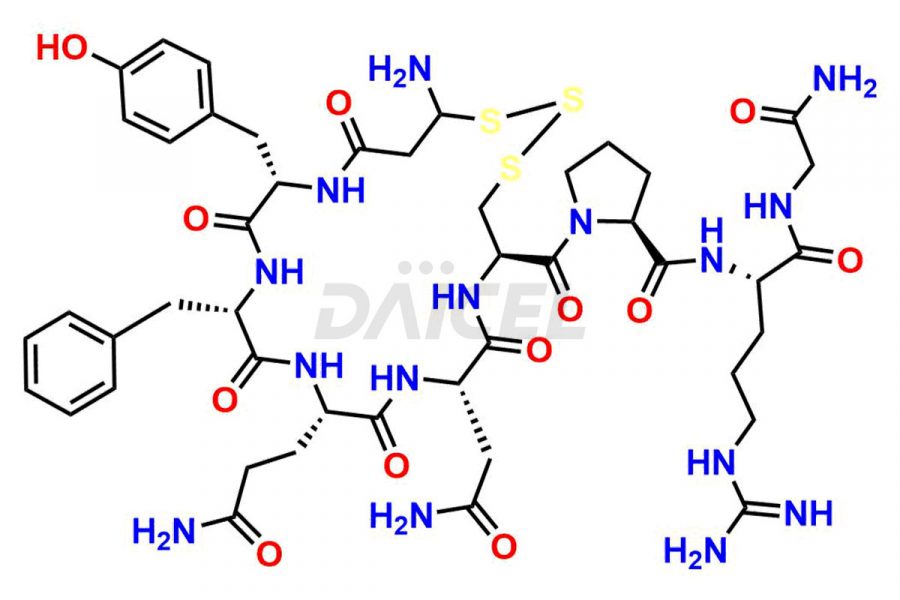
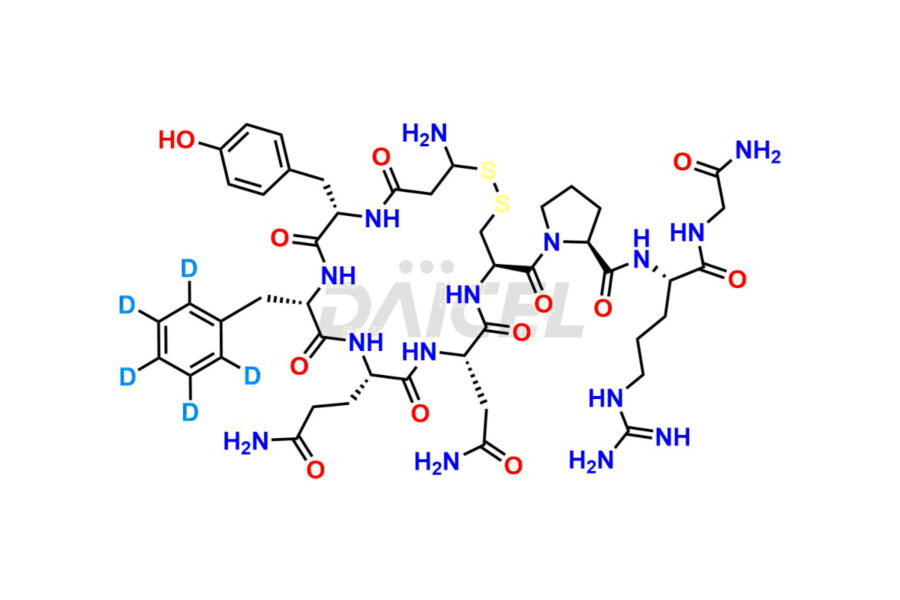
Daicel Pharma synthesizes more than twenty-five high-quality Vasopressin impurities such as Ac-Asp (5)-Vasopressin, Anti-Parallel Dimer-Vasopressin, Asp5- Vasopressin, D-Arg(8)-Vasopressin, Di-sulfonic acid- Vasopressin, Parallel Dimer–Vasopressin, Trisulfide-Vasopressin, Vasopressin D5, β-Asp (5)-Vasopressin, and more. The impurities are crucial in determining the quality, stability, and biological safety of the active pharmaceutical ingredient (API), Vasopressin. Moreover, Daicel Pharma offers custom synthesis of Vasopressin impurities and delivers them globally.
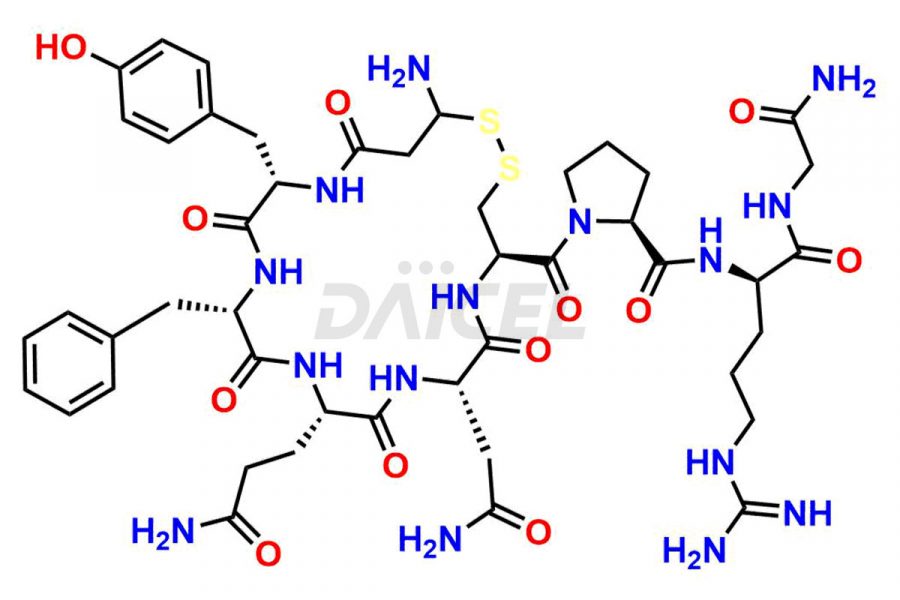
Vasopressin [CAS: 11000-17-2] is a small nine-amino acid peptide hormone secreted from the posterior pituitary gland. The hormone has important vasopressor and antidiuretic effects and is also involved in glycogenolysis.
Vasopressin: Use and Commercial Availability
Vasopressin is a synthetic peptide hormone that shares the same properties as the natural hormone, Antidiuretic Hormone (ADH) or Arginine Vasopressin (AVP). It regulates fluid levels in the body by controlling water reabsorption in the kidneys. It is produced in the hypothalamus and stored in the posterior pituitary gland. Vasopressin has antidiuretic effects and can cause vasoconstriction, which increases blood pressure and systemic vascular resistance.
Vasopressin is available as an injection. It’s marketed under various names, depending on the country and manufacturer, including Vasostrict, Reverin, Pitressin, ADH, Argipressin, Empressin, etc.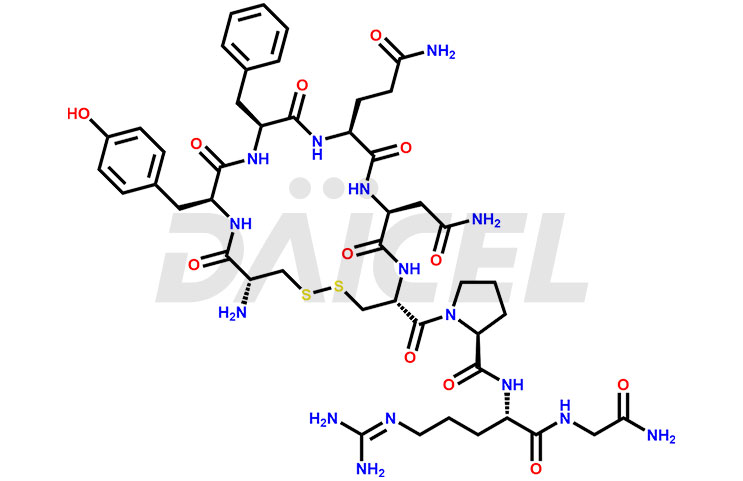
Vasopressin Structure and Mechanism of Action
The chemical formula for Vasopressin is C46H65N15O12S2, and its molecular weight is approximately 1084.2 g/mol.
Vasopressin binds to vascular V1 receptors. The Vascular V1 receptors are directly coupled to phospholipase C, resulting in calcium release leading to vasoconstriction. Vasopressin also stimulates antidiuresis.
Vasopressin Impurities and Synthesis
Ensuring the safety and effectiveness of drugs is essential in pharmaceutical quality control. Impurities that arise during synthesis1 and storage of arginine Vasopressin (AVP) can have unexpected effects on the drug’s quality, safety, and efficacy. Advanced analytical methods such as liquid chromatography/high-resolution mass spectrometry (LC/HRMS) and HPLC are employed to detect and measure these impurities. This method is valuable for identifying related peptide impurities in Vasopressin, allowing for precise identification and quantification of any potential impurities that may affect the drug’s quality.
Daicel offers a Certificate of Analysis (CoA) from a cGMP-compliant analytical facility for more than twenty-five Vasopressin impurities, including Ac-Asp (5)-Vasopressin, Anti-Parallel Dimer-Vasopressin, Asp5- Vasopressin, D-Arg(8)-Vasopressin, Di-sulfonic acid- Vasopressin, Parallel Dimer–Vasopressin, Trisulfide-Vasopressin, Vasopressin D5, β-Asp (5)-Vasopressin, and more. The CoA includes complete characterization data, such as 1H NMR, 13C NMR, IR, MASS, and HPLC3 purity. We also provide Amino Acid composition, Sequence analysis and CHN on request. We also give a complete characterization report on delivery. Daicel has the technology and expertise to prepare any unknown Vasopressin impurity or degradation product. We also provide labeled compounds to quantify the efficacy of generic Vasopressin. Daicel offers highly pure Vasopressin D5, a deuterium-labeled standard of Vasopressin for bioanalytical research and BA/BE studies with isotope data in CoA.