General Information
Siponimod Impurities and Siponimod
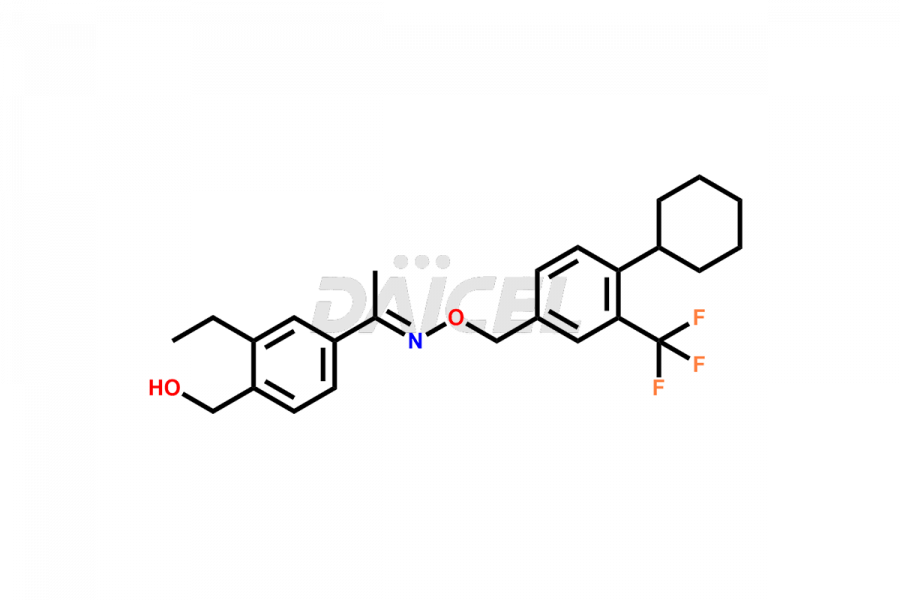
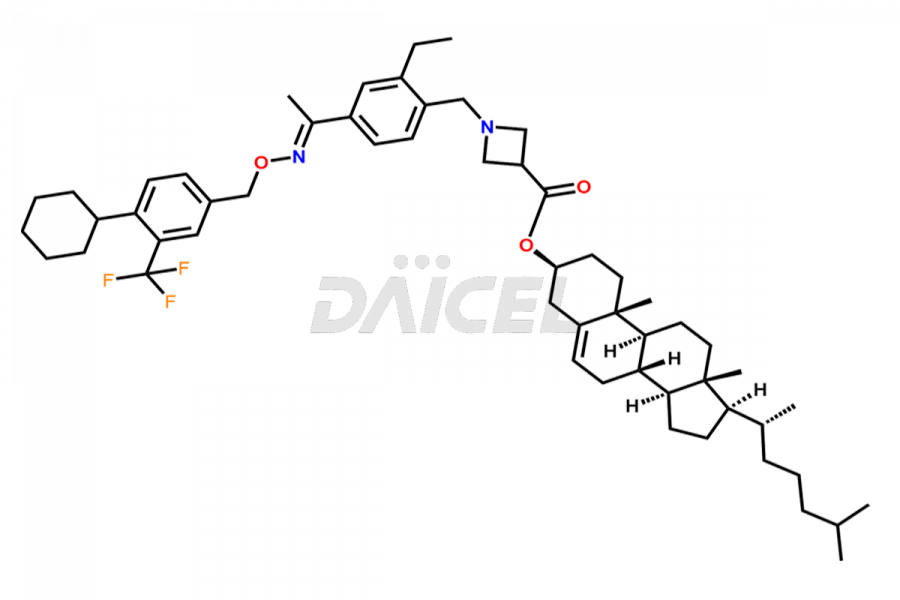
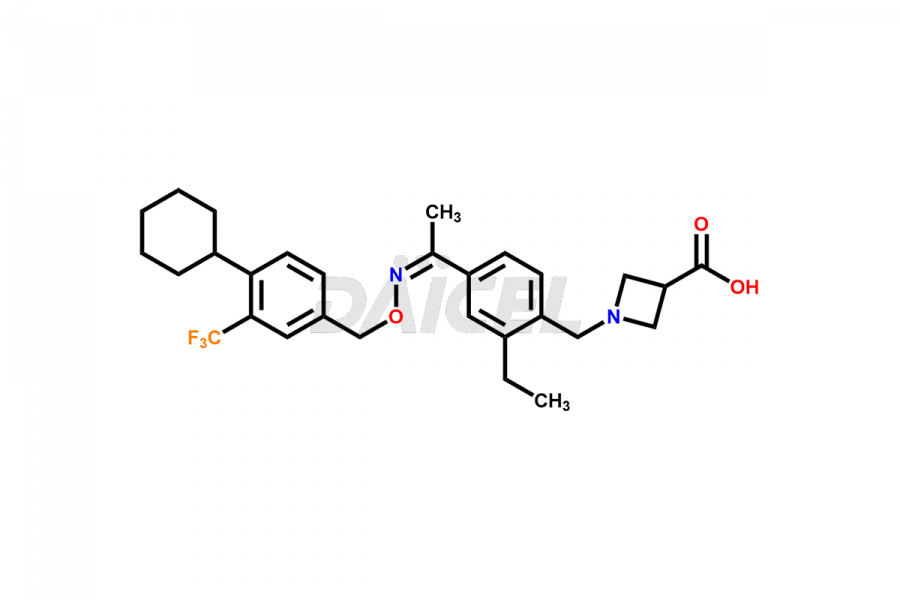
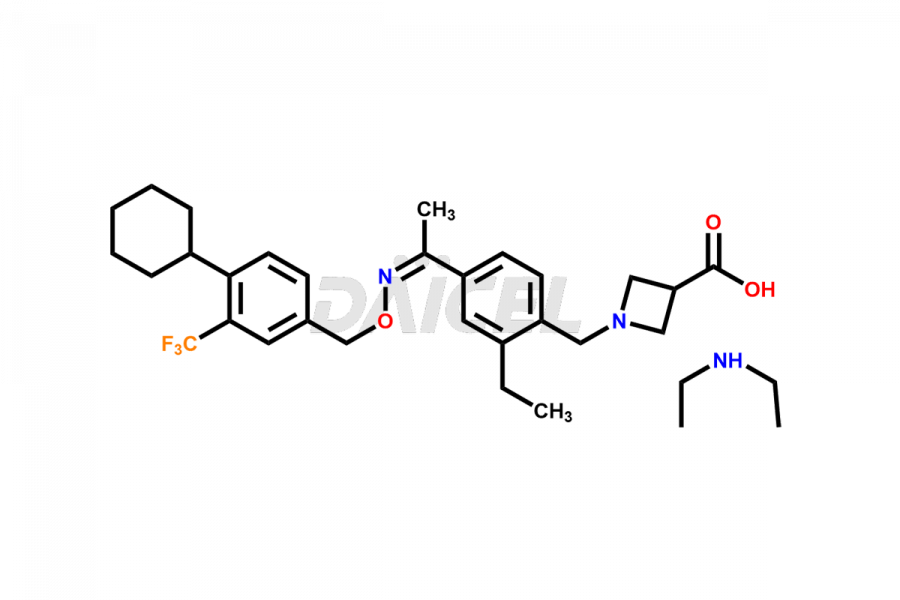
Daicel Pharma is a reliable partner for synthesizing high-quality Siponimod impurities, specifically 1-(4-ethyl-3-(hydroxymethyl)phenyl)ethan-1-one, Siponimod M17, and Siponimod Z-Isomer diethylamine salt. These impurities help assess the quality, stability, and safety of the active pharmaceutical ingredient, Siponimod. Daicel Pharma also offers custom synthesis of Siponimod impurities, which can be shipped worldwide to meet customers’ unique requirements.
Siponimod [CAS: 1230487-00-9] is a medication that modulates a type of receptor called sphingosine 1-phosphate (S1P). It treats relapsing forms of multiple sclerosis (MS), including clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease.
Siponimod: Use and Commercial Availability
Siponimod is available as Mayzent, an oral medication to treat relapsing forms of multiple sclerosis (MS) in adults. It is a newly approved drug by the US FDA and Health Canada. Siponimod is an immunomodulatory agent treating patients with multiple sclerosis.
Siponimod Structure and Mechanism of Action 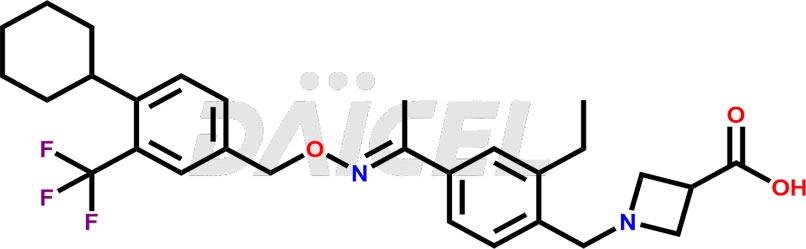
The chemical name of Siponimod is 1-[[4-[(1E)-1-[[[4-Cyclohexyl-3-(trifluoromethyl)phenyl]methoxy]imino]ethyl]-2-ethylphenyl]methyl]-3-azetidinecarboxylic acid. Its chemical formula is C29H35F3N2O3, and its molecular weight is approximately 516.6 g/mol.
Siponimod binds with sphingosine-1-phosphate (SIP) receptors 1 and 5 with affinity. It reduces lymphocyte migration into the central nervous system.
Siponimod Impurities and Synthesis
The impurities in Siponimod form during various stages of the manufacturing process, including synthesis1, purification, and storage. It is essential to identify and control impurities to ensure the safety and efficacy of the final product.
Daicel Pharma issues a Certificate of Analysis (CoA) for Siponimod impurity standards, including 1-(4-ethyl-3-(hydroxymethyl)phenyl)ethan-1-one, Siponimod M17, and Siponimod Z-Isomer diethylamine salt. The CoA is from an analytical facility that complies with current Good Manufacturing Practices (cGMP) and includes comprehensive characterization data, such as 1H NMR, 13C NMR, IR, MASS, and HPLC purity2. We give additional characterization data such as 13C-DEPT and CHN on request. Daicel Pharma can prepare unknown Siponimod impurities or degradation products. A complete characterization report accompanies each delivery.