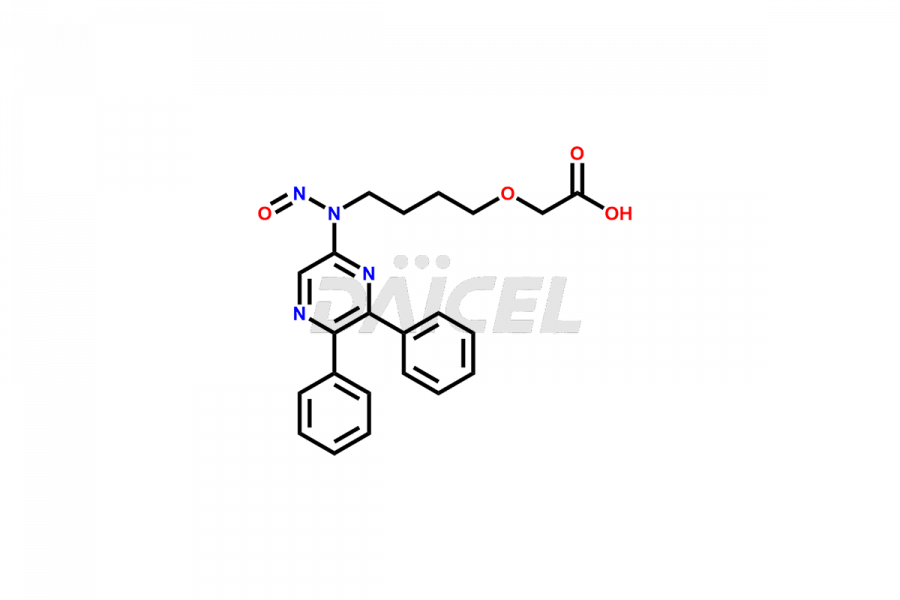
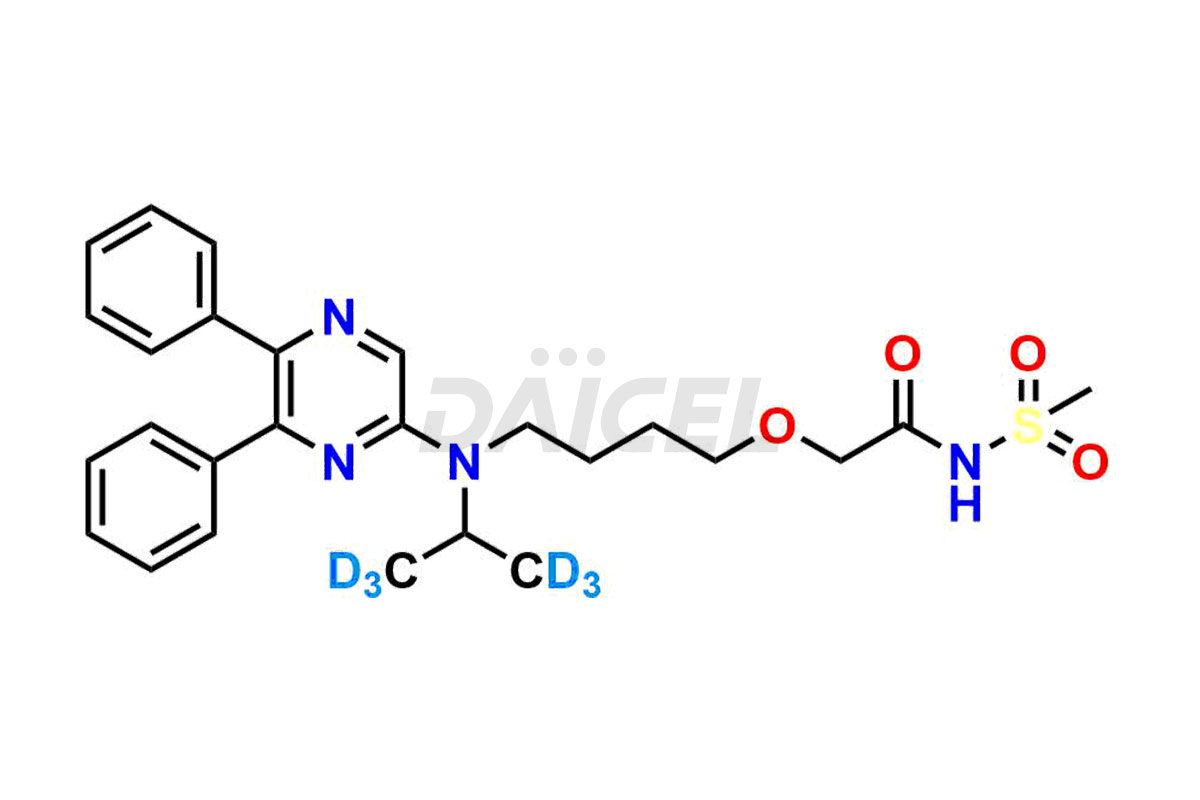
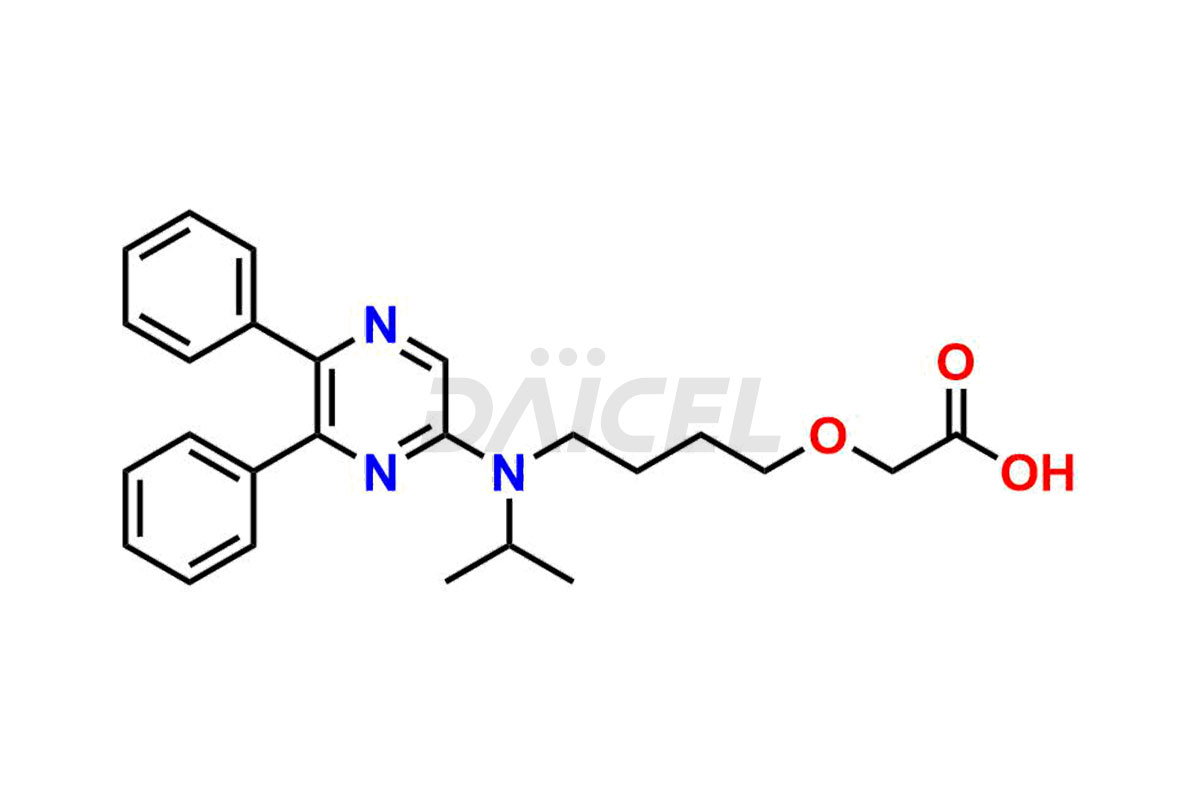
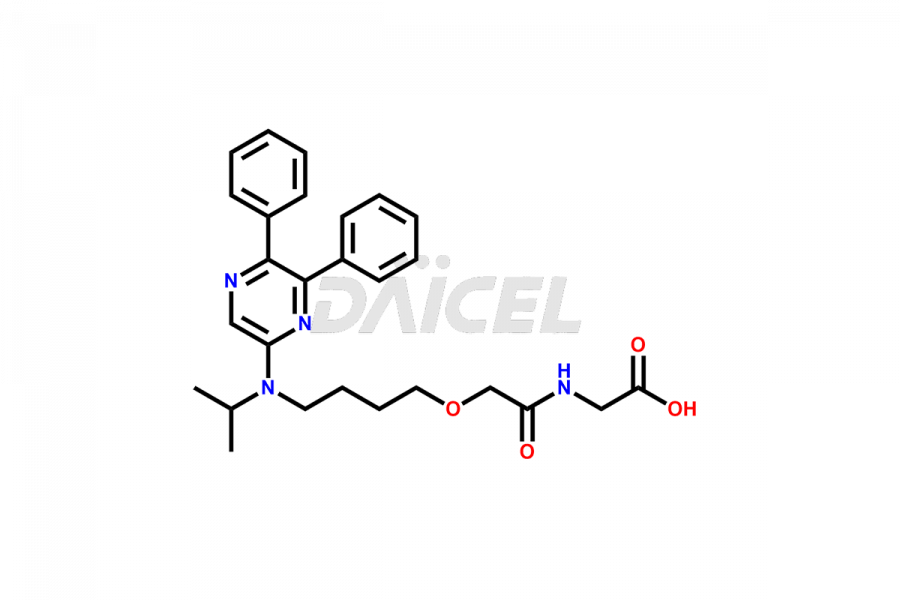
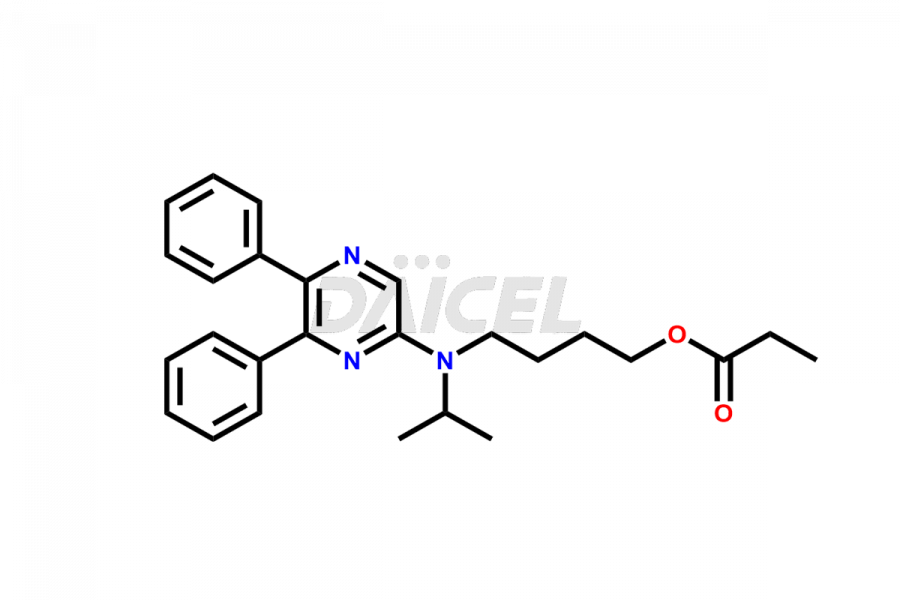
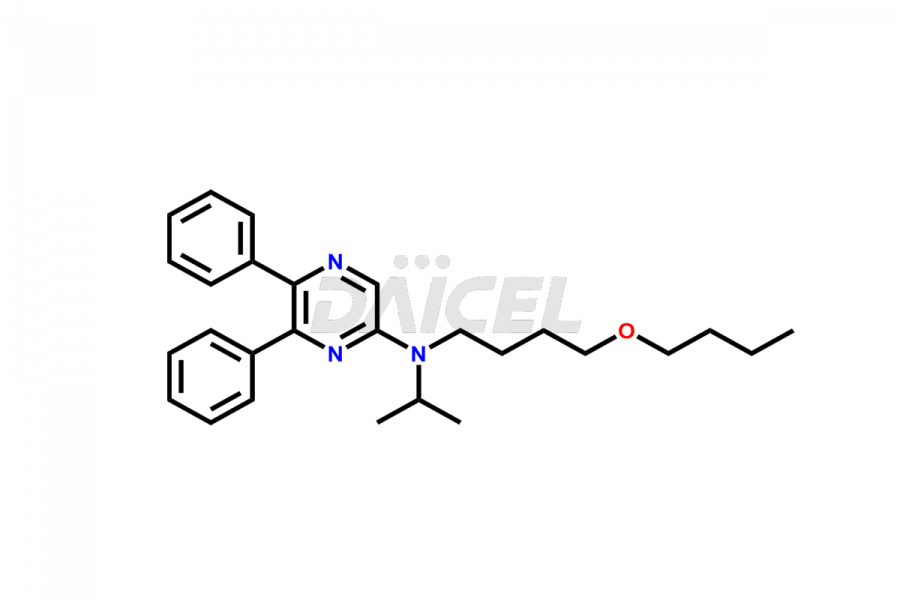
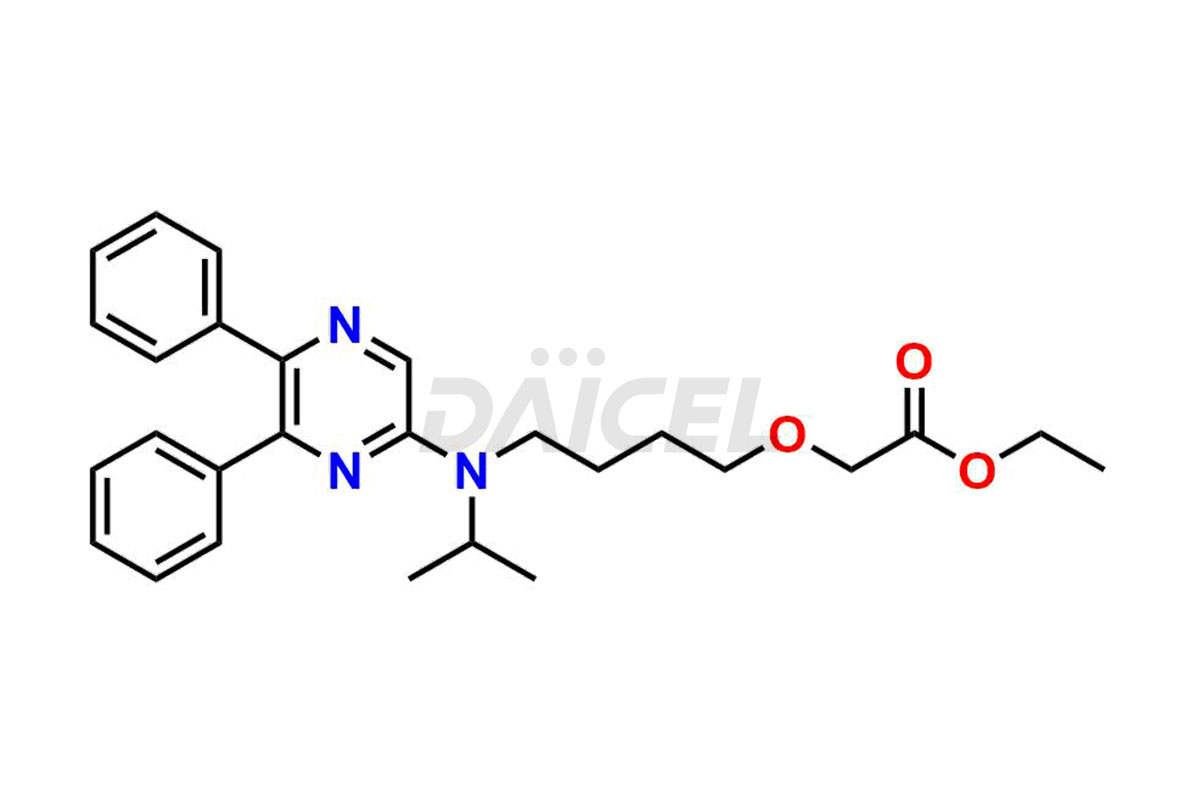
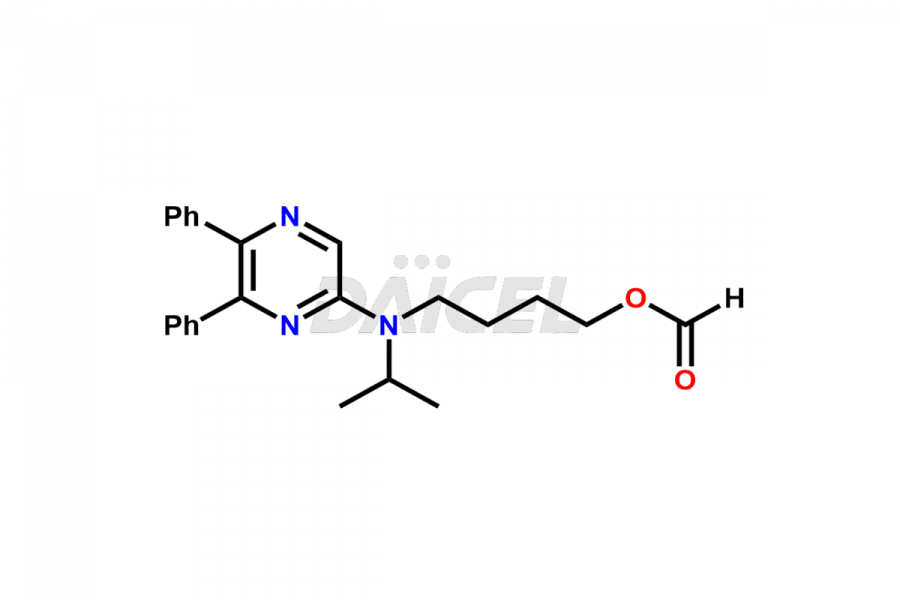
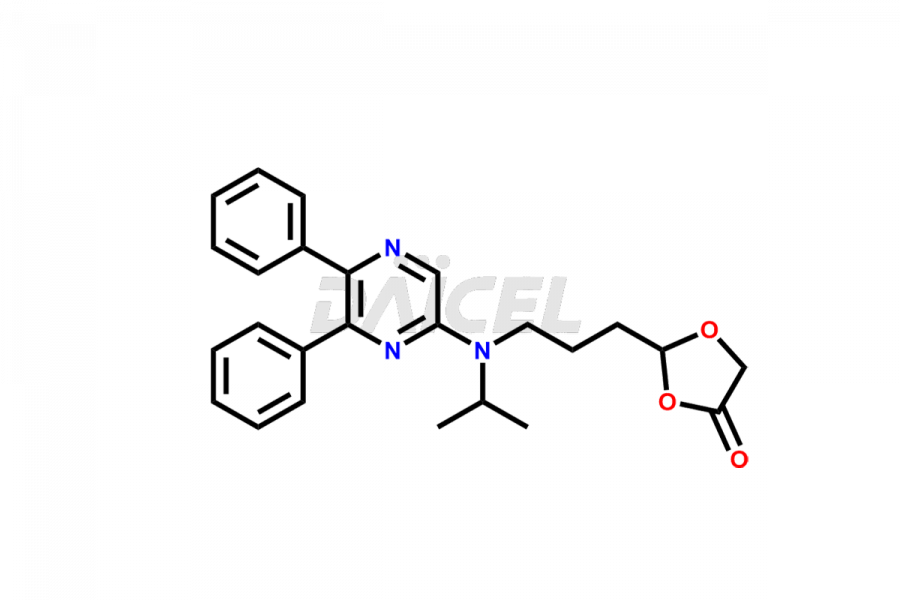
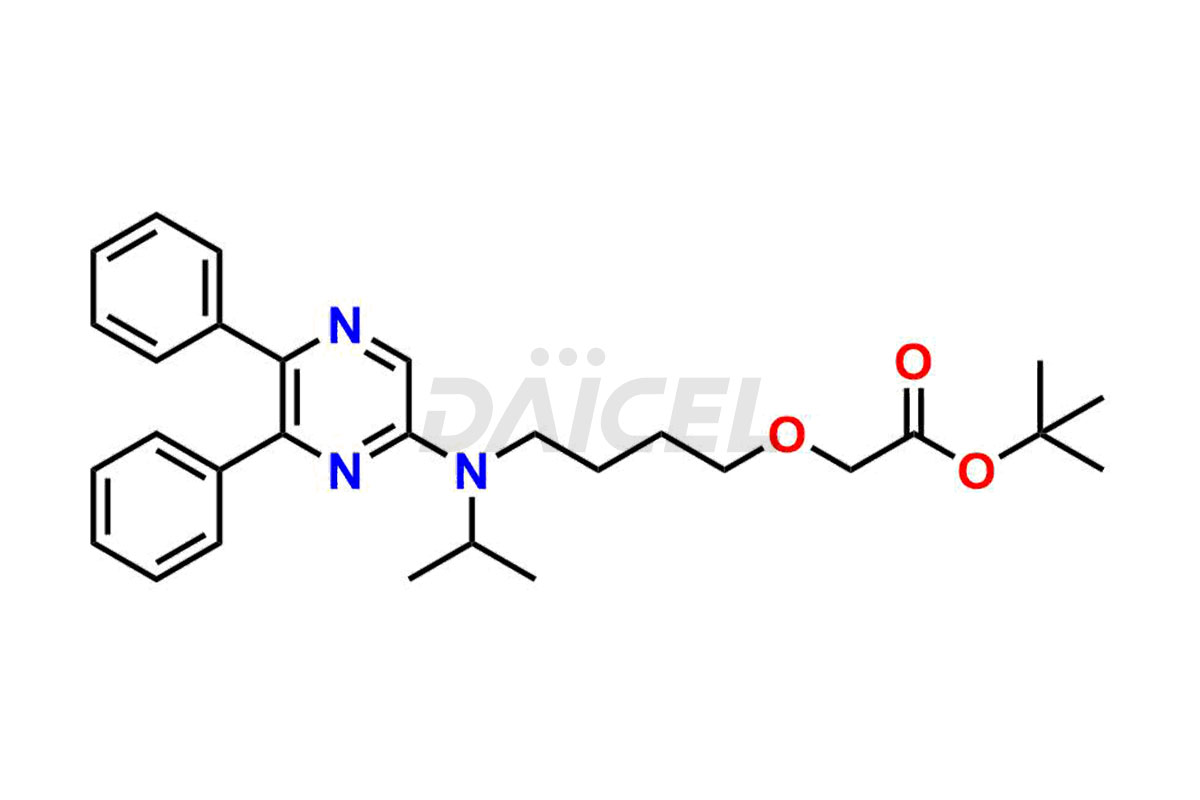
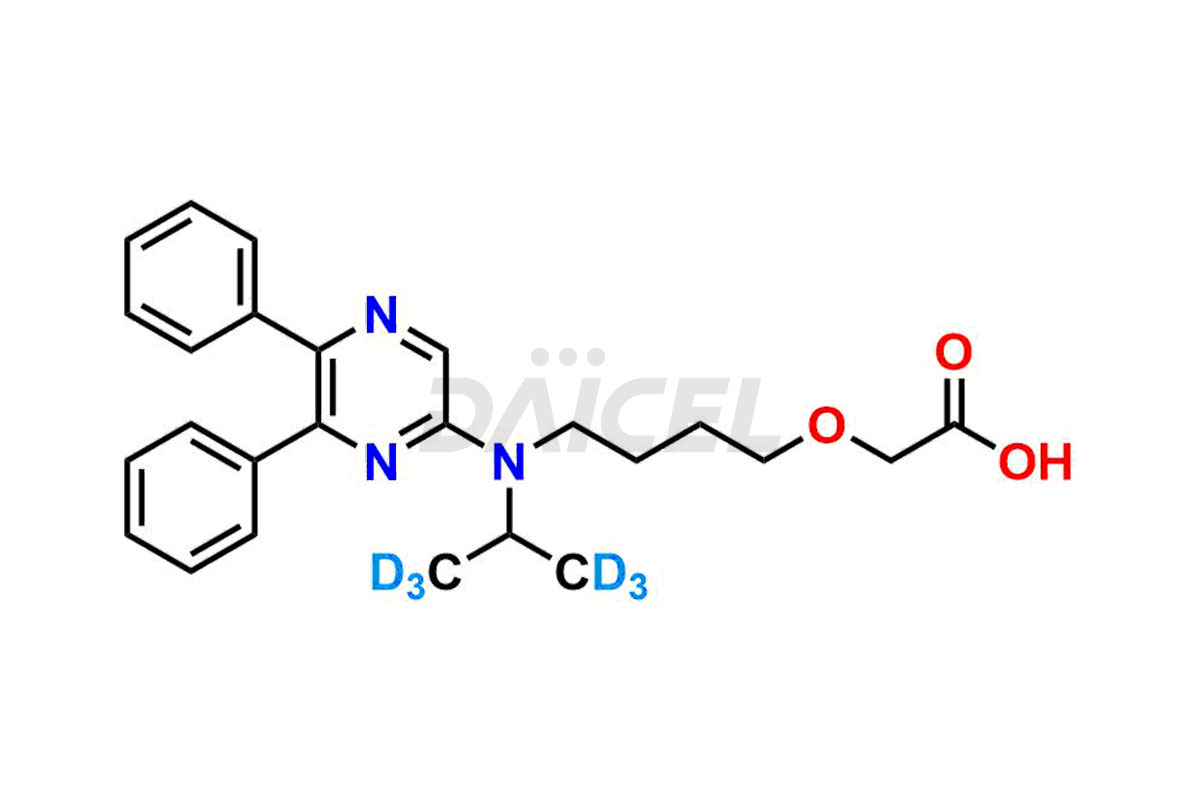
General Information
Selexipag Impurities and Selexipag
Daicel Pharma is a reliable source for synthesizing high-quality Selexipag impurities, like Selexipag Active metabolite, Selexipag Impurity 1, Selexipag Impurity 8, and many more. Daicel Pharma offers a range of Selexipag impurities that critically assess active pharmaceutical ingredients’ quality, stability, and safety. Additionally, Daicel Pharma specializes in custom synthesis of Selexipag impurities that meet client requirements. These high-quality impurities can be shipped worldwide, providing flexibility and convenience to customers.
Selexipag [CAS: 475086-01-2] is a member of the class of pyrazine drugs that treat pulmonary arterial hypertension (PAH), delay disease progression, and reduce the risk of hospitalization.
Selexipag: Use and Commercial Availability
Selexipag is a medication to treat pulmonary arterial hypertension (PAH). Selexipag belongs to a class of drugs called prostacyclin receptor agonists, which work by dilating (widening) the blood vessels in the lungs, reducing the workload on the heart, and improving blood flow. Selexipag delays disease progression and improves survival in patients.
Selexipag is available under Uptravi, which contains the active ingredient, Selexipag.
Selexipag Structure and Mechanism of Action 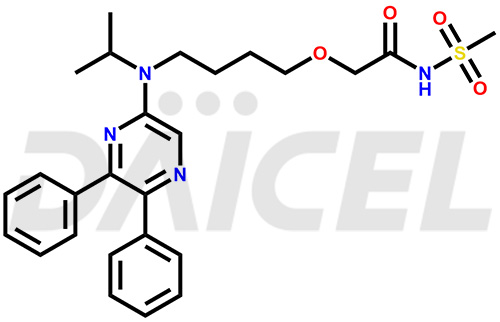
The chemical name of Selexipag is 2-[4-[(5,6-Diphenyl-2-pyrazinyl)(1-methylethyl)amino]butoxy]-N-(methylsulfonyl)acetamide. Its chemical formula is C26H32N4O4S, and its molecular weight is approximately 496.6 g/mol.
Selexipag is a prostacyclin receptor agonist. It hydrolyzes to its potent active metabolite using y carboxylesterase 1.
Selexipag Impurities and Synthesis
During the manufacturing process1 of Selexipag, impurities may form, potentially reducing its effectiveness. They can originate from various sources, including raw materials, intermediates, and chemicals used to synthesize Selexipag. It is necessary to manage and monitor these impurities closely to ensure the drug’s effectiveness and safety.
Daicel provides a comprehensive Certificate of Analysis (CoA) for Selexipag impurity standards, including Selexipag Active metabolite, Selexipag Impurity 1, Selexipag Impurity 8, and more. The CoA includes detailed characterization data such as 1H NMR, 13C NMR, IR, MASS, and HPLC purity2. Additionally, upon delivery, we give a complete 13C-DEPT. Daicel Pharma possesses the technology and expertise to synthesize any unknown Selexipag impurity or degradation product. We also offer labeled compounds. For bioanalytical research and BA/BE studies, Daicel Pharma supplies Selexipag – D6 and Selexipag Metabolite – D6, deuterium-labeled standards of Selexipag.