General Information
Palbociclib Impurities and Palbociclib
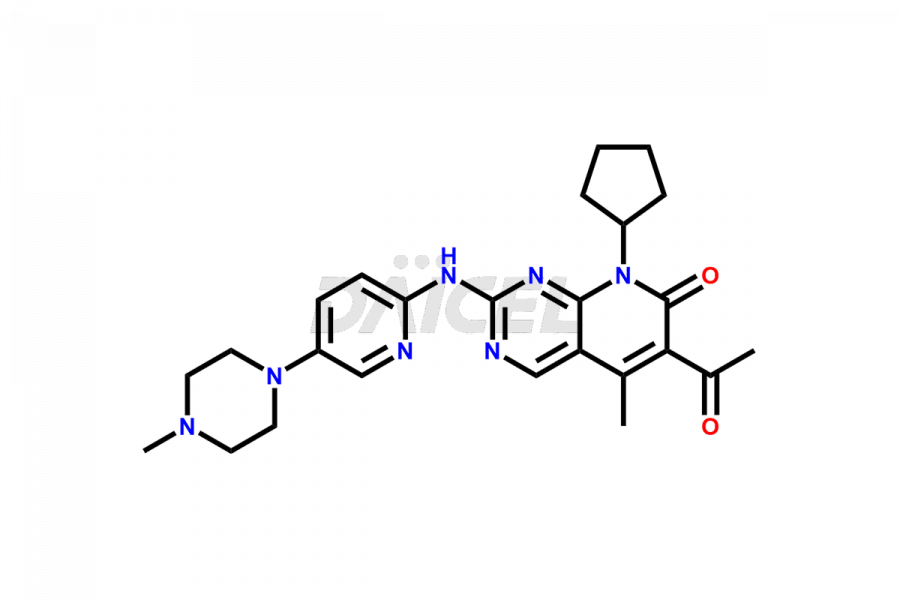
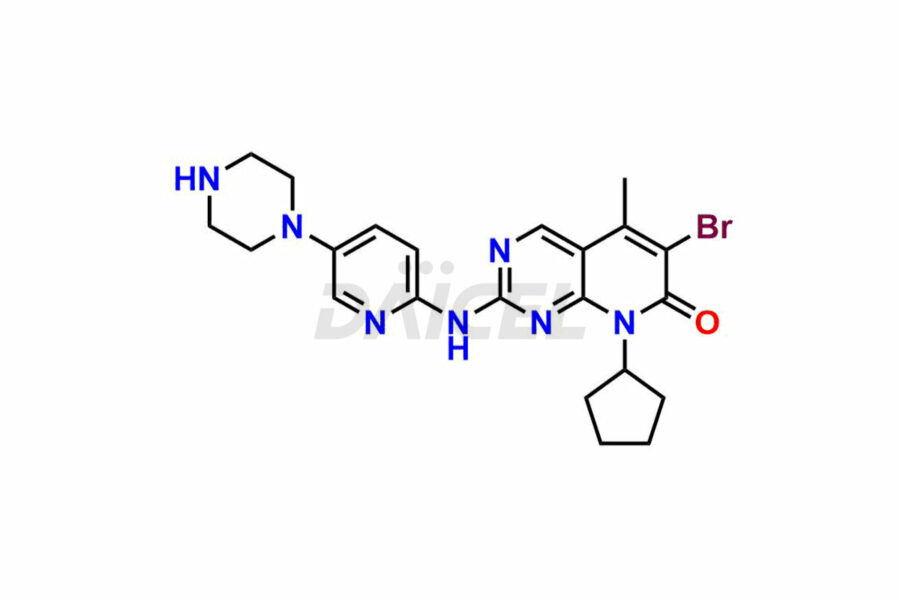
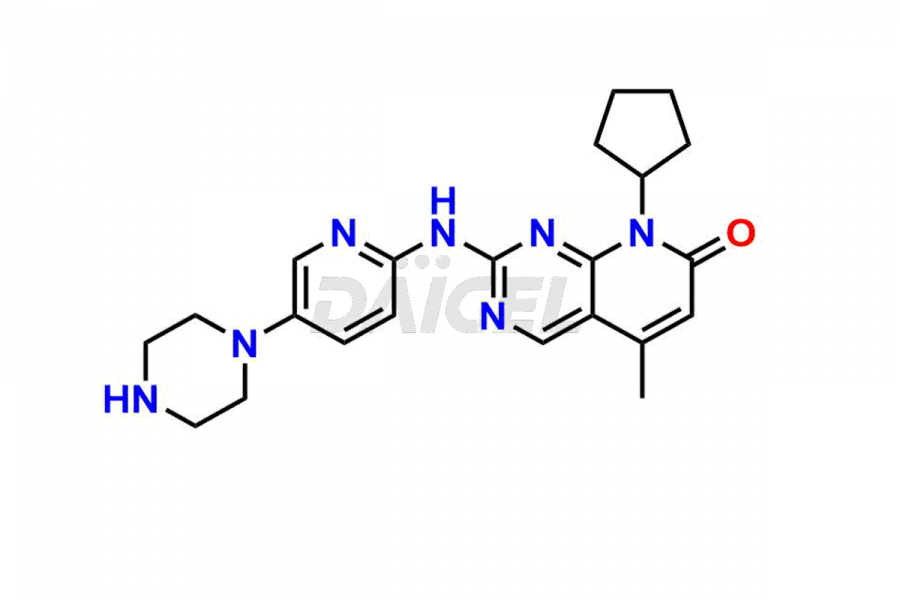
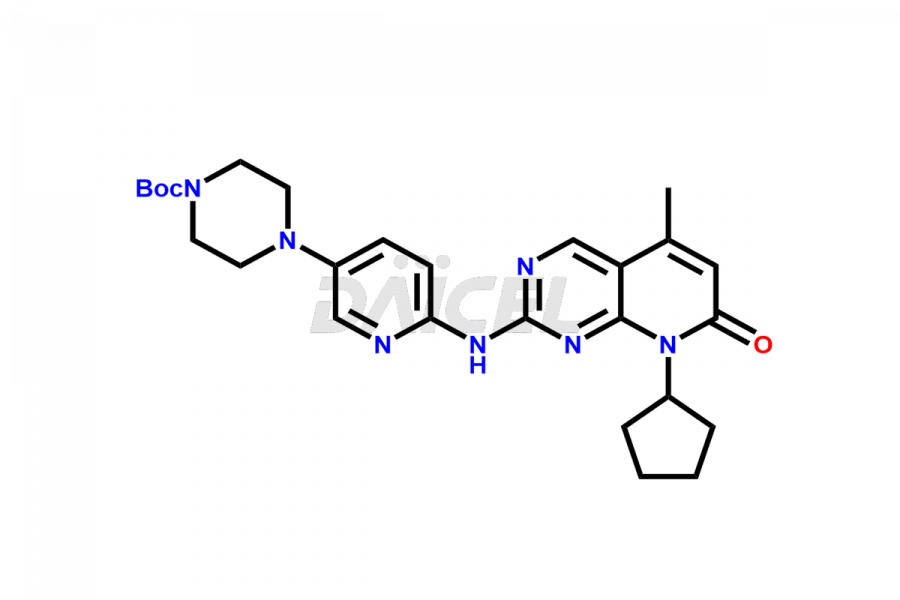
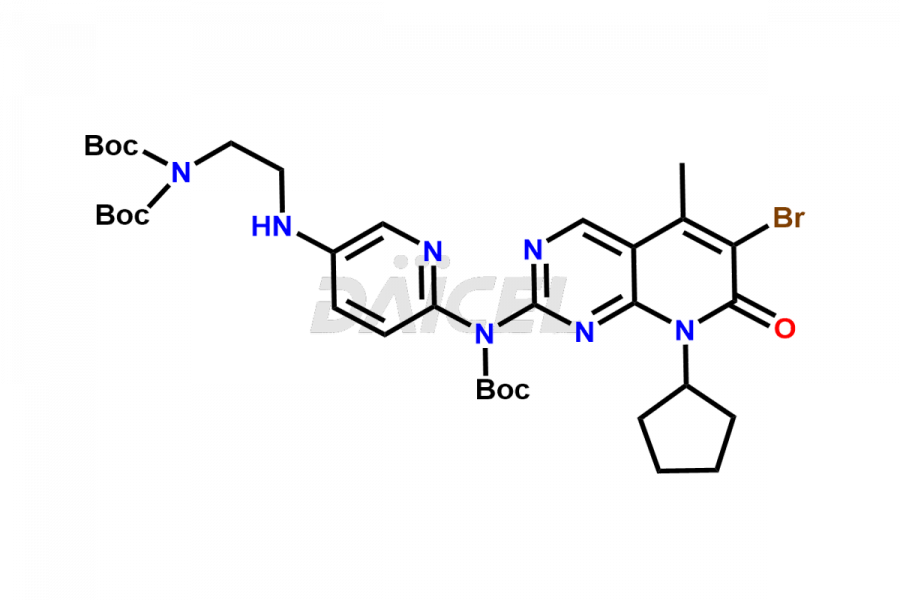
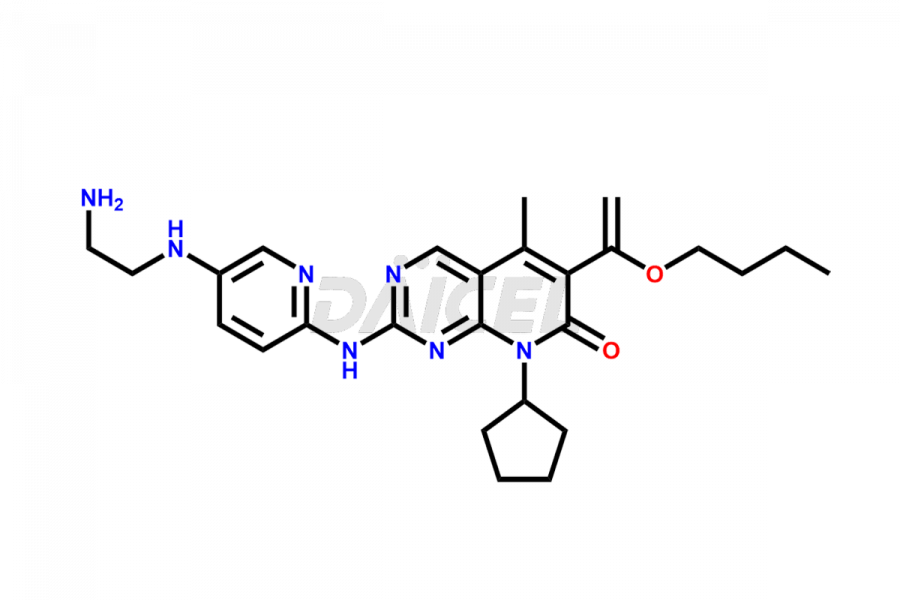
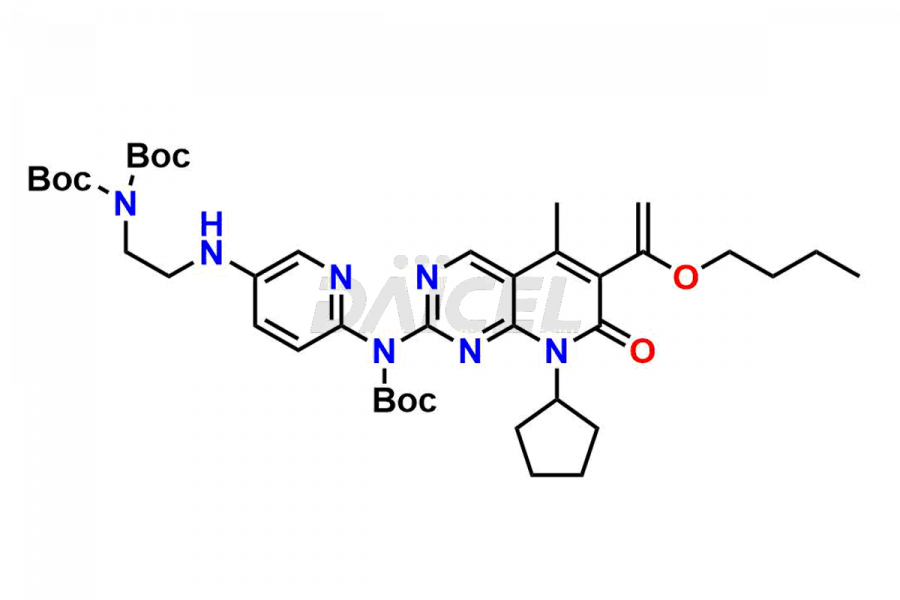
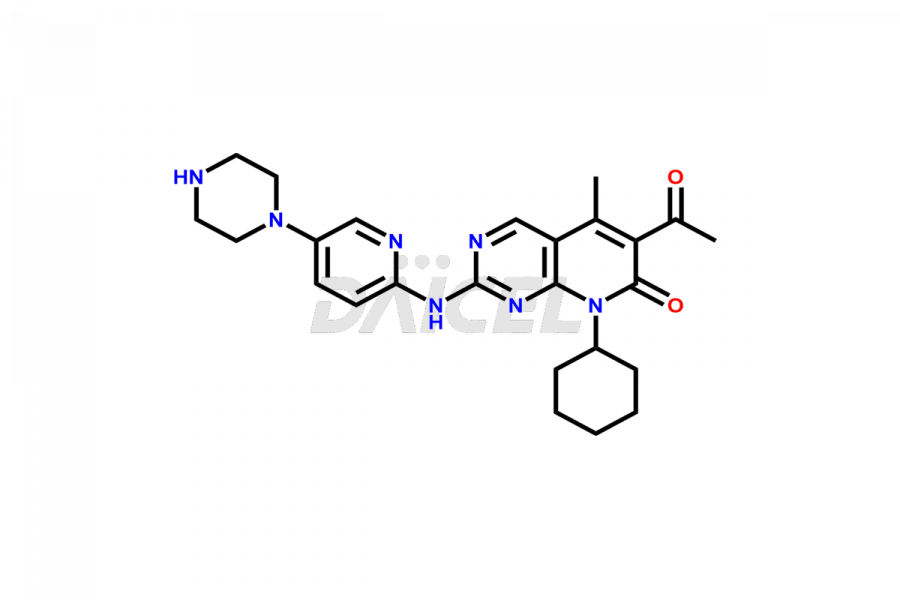
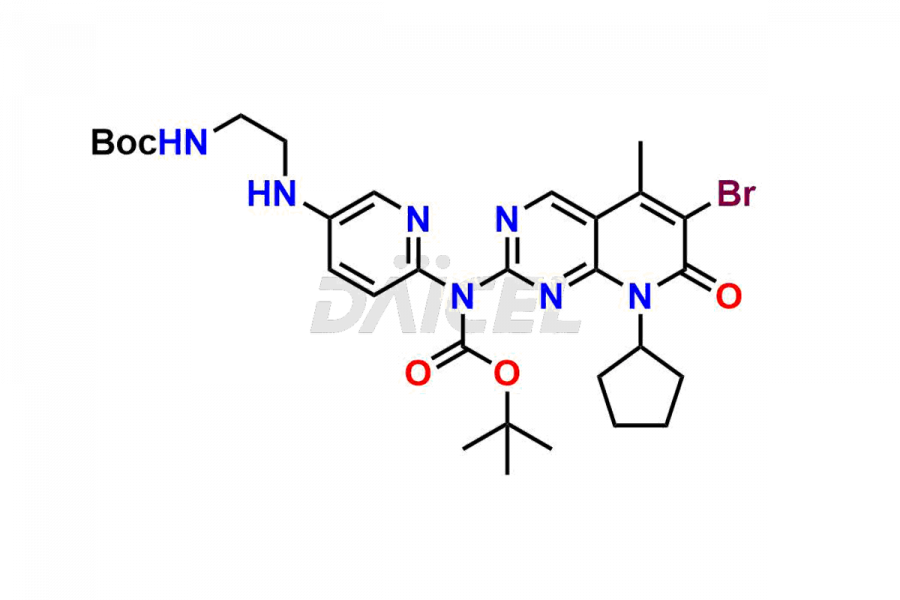
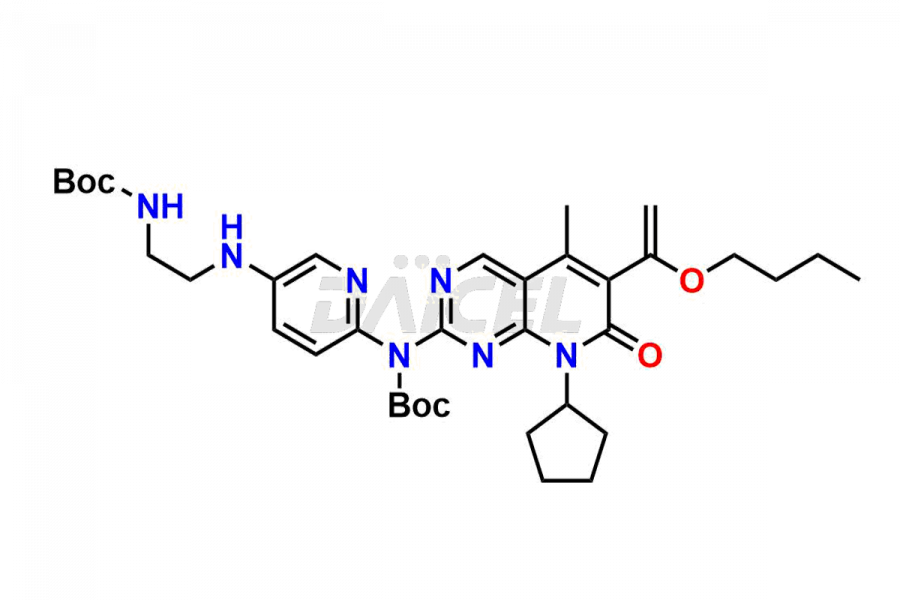
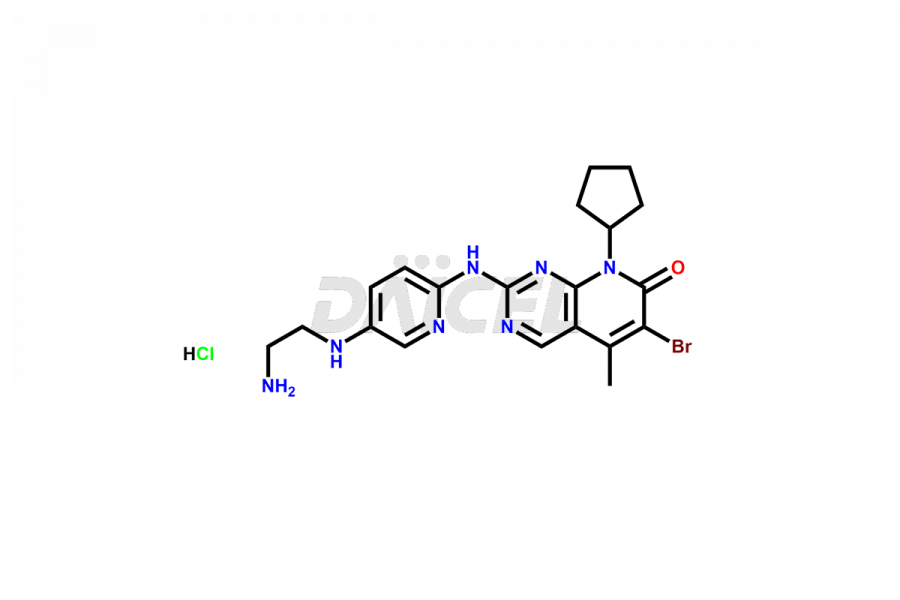
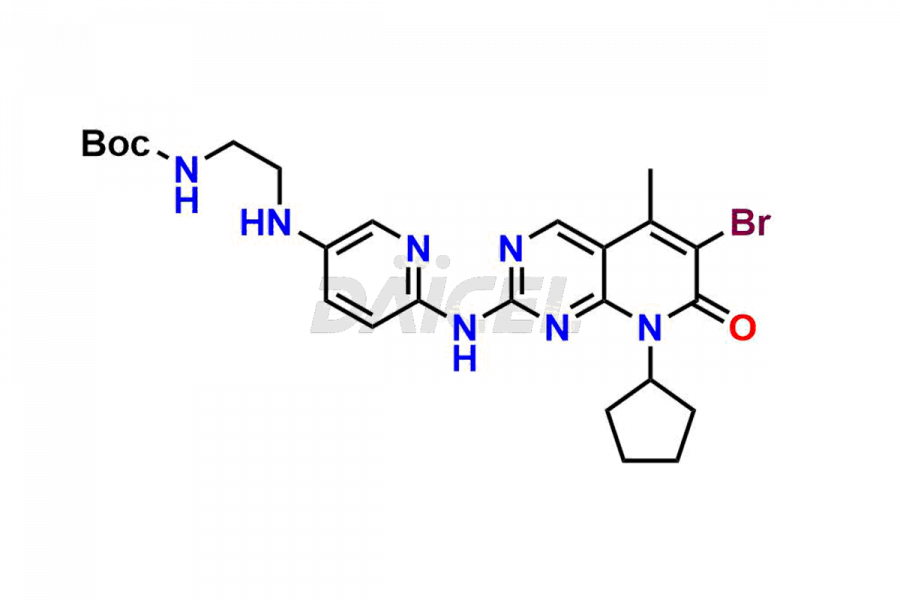
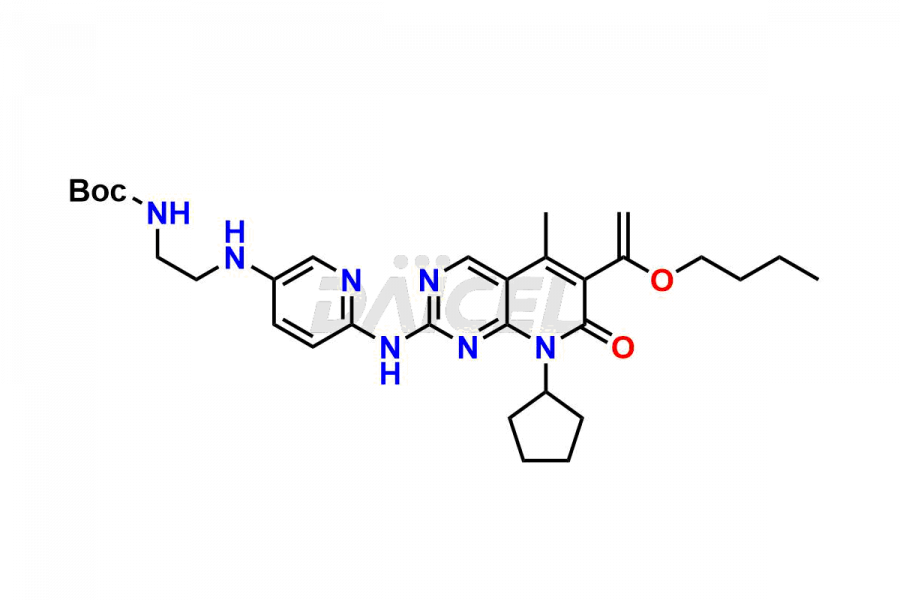
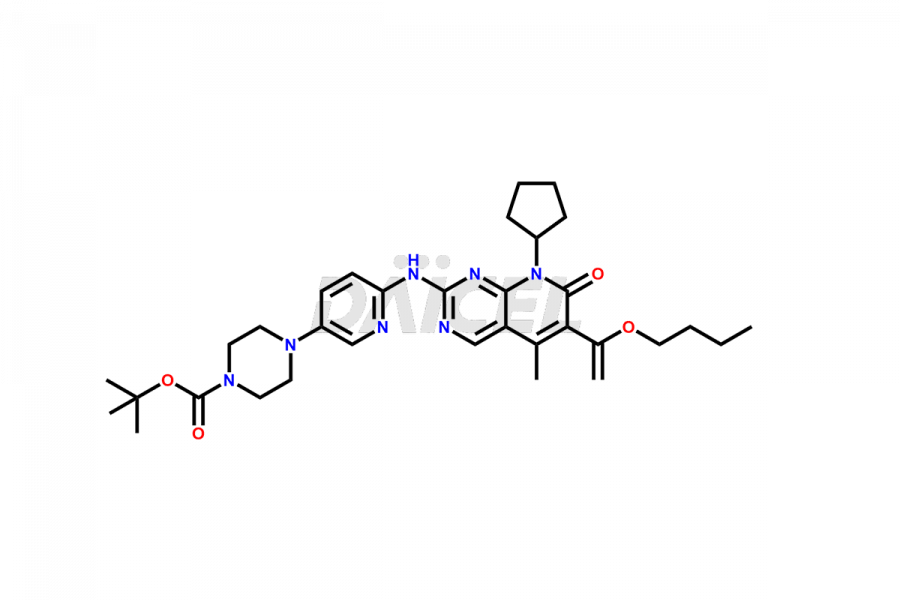
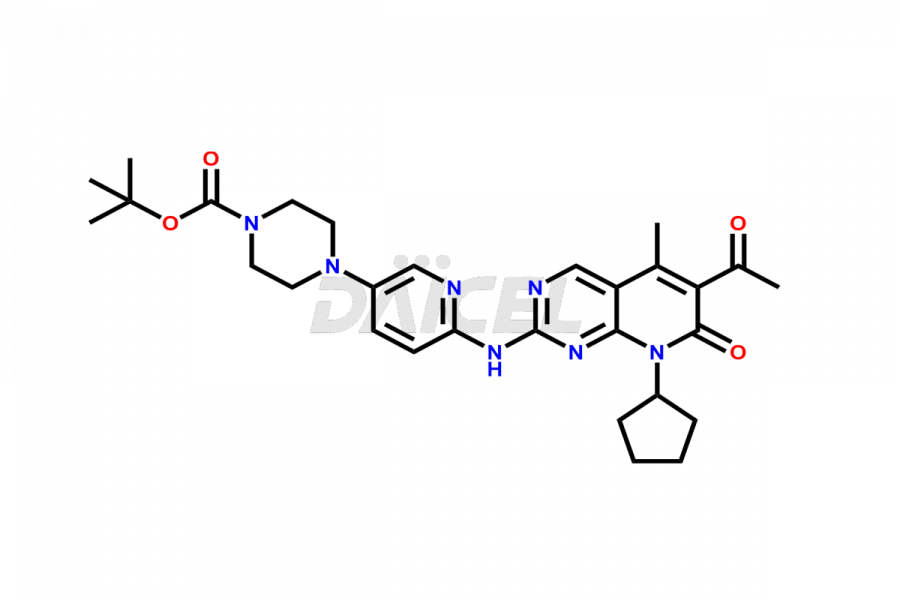
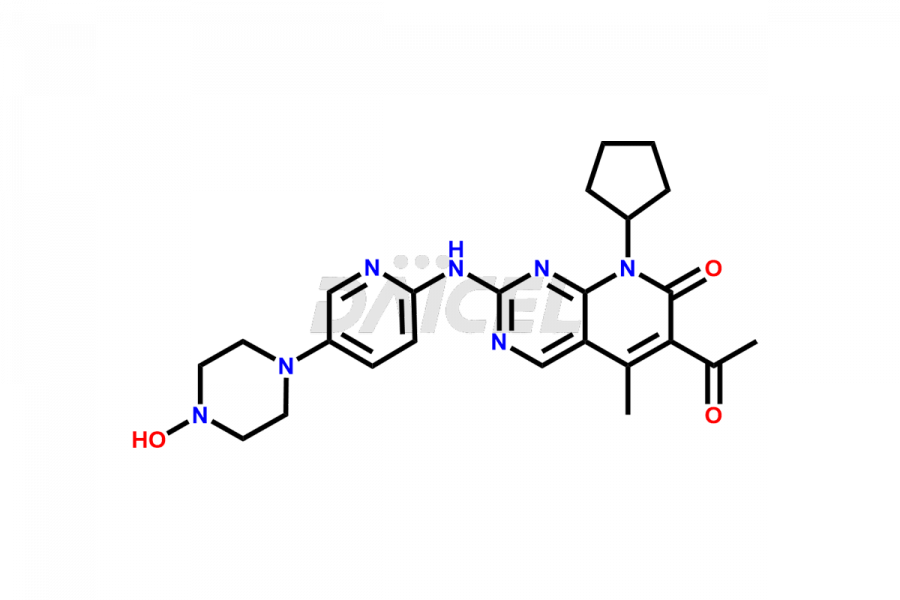
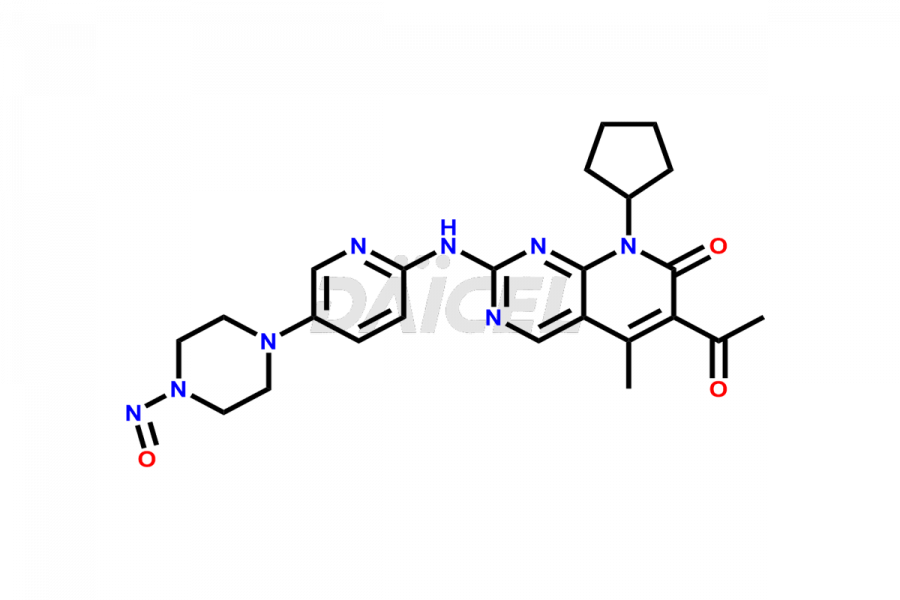
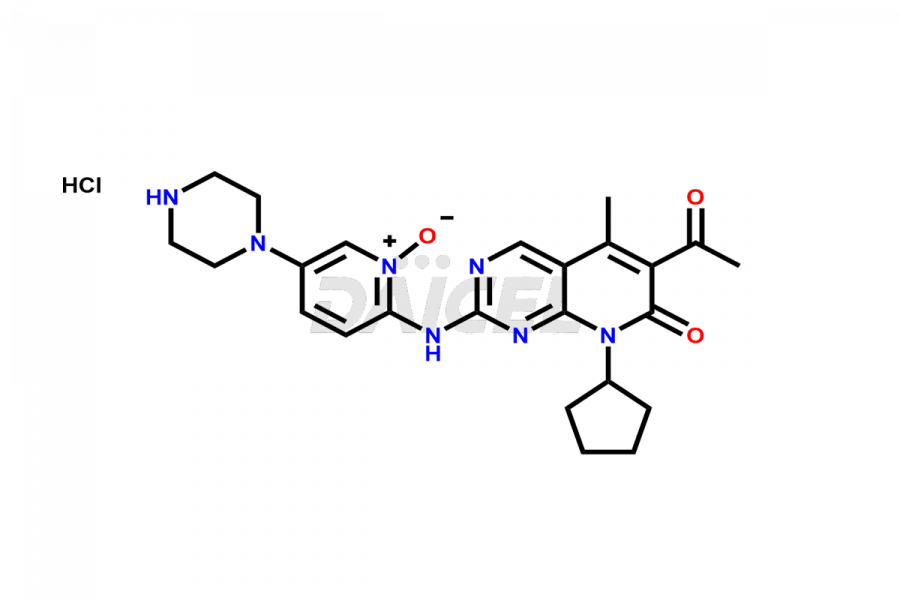
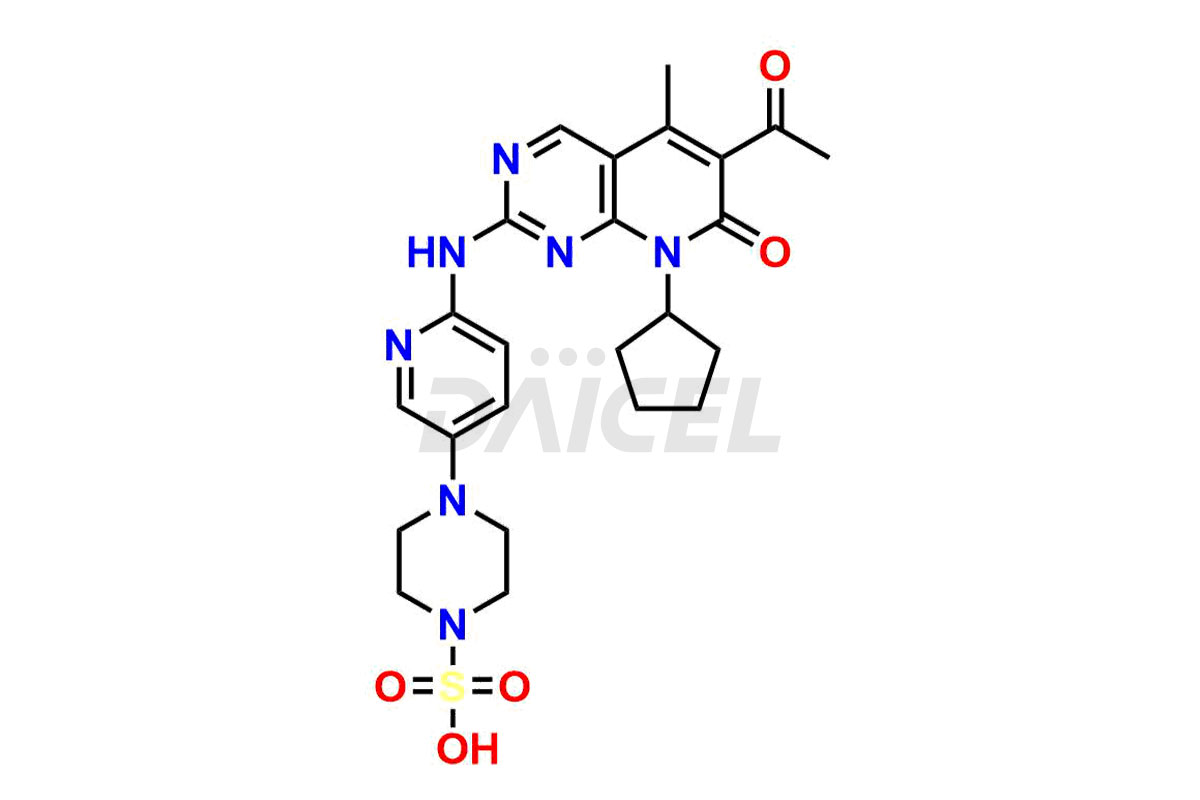
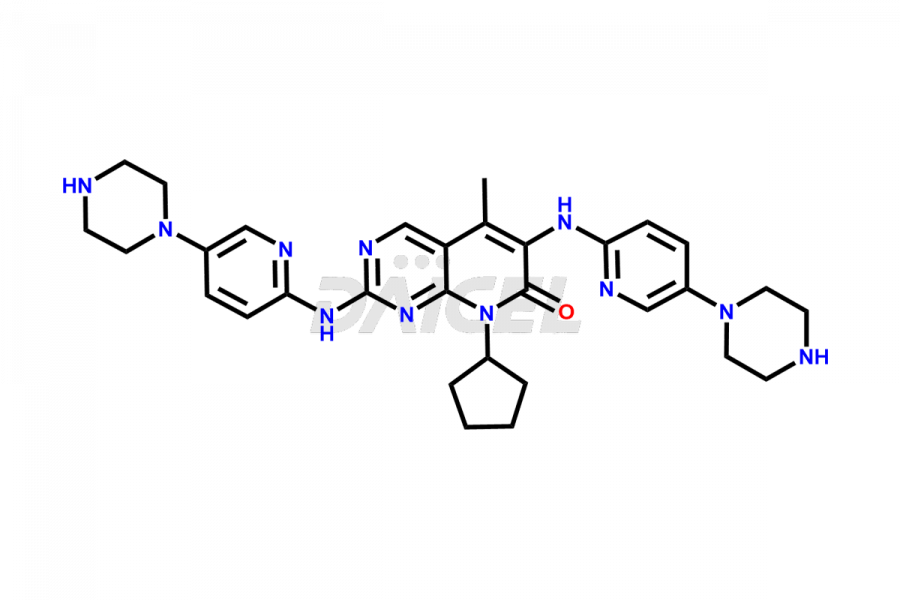
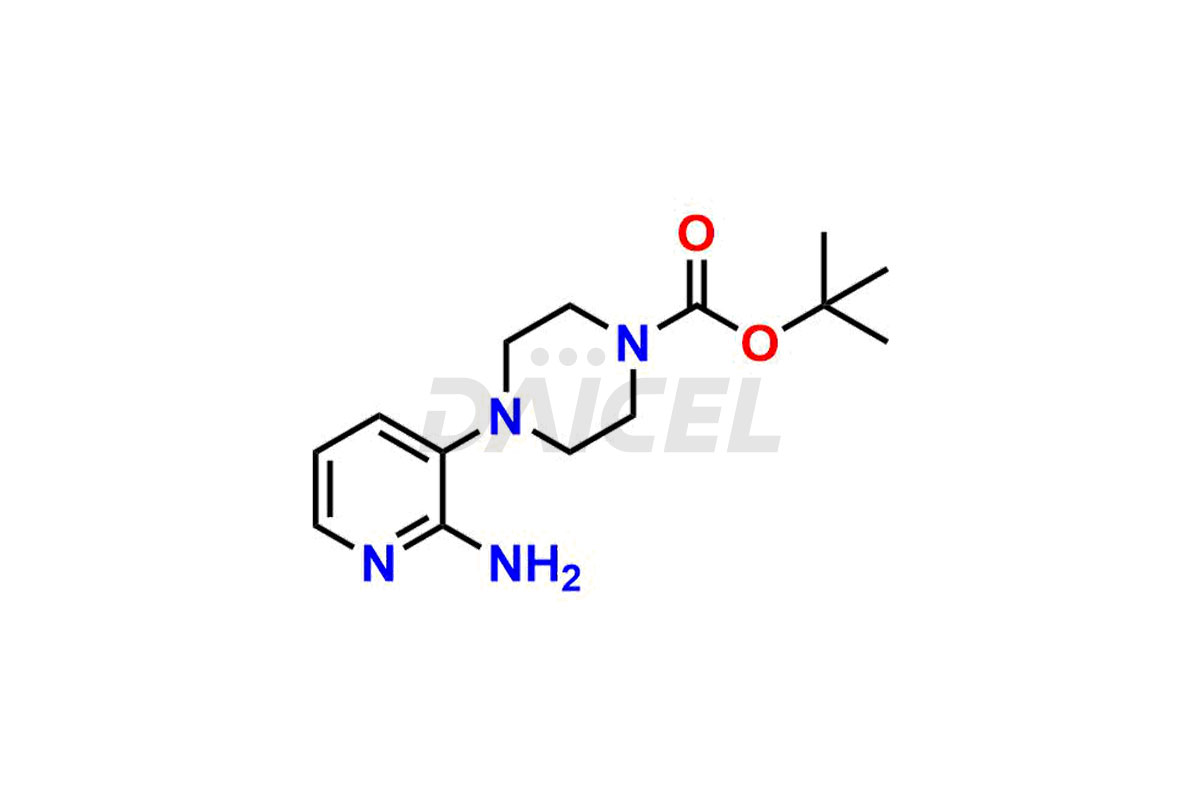
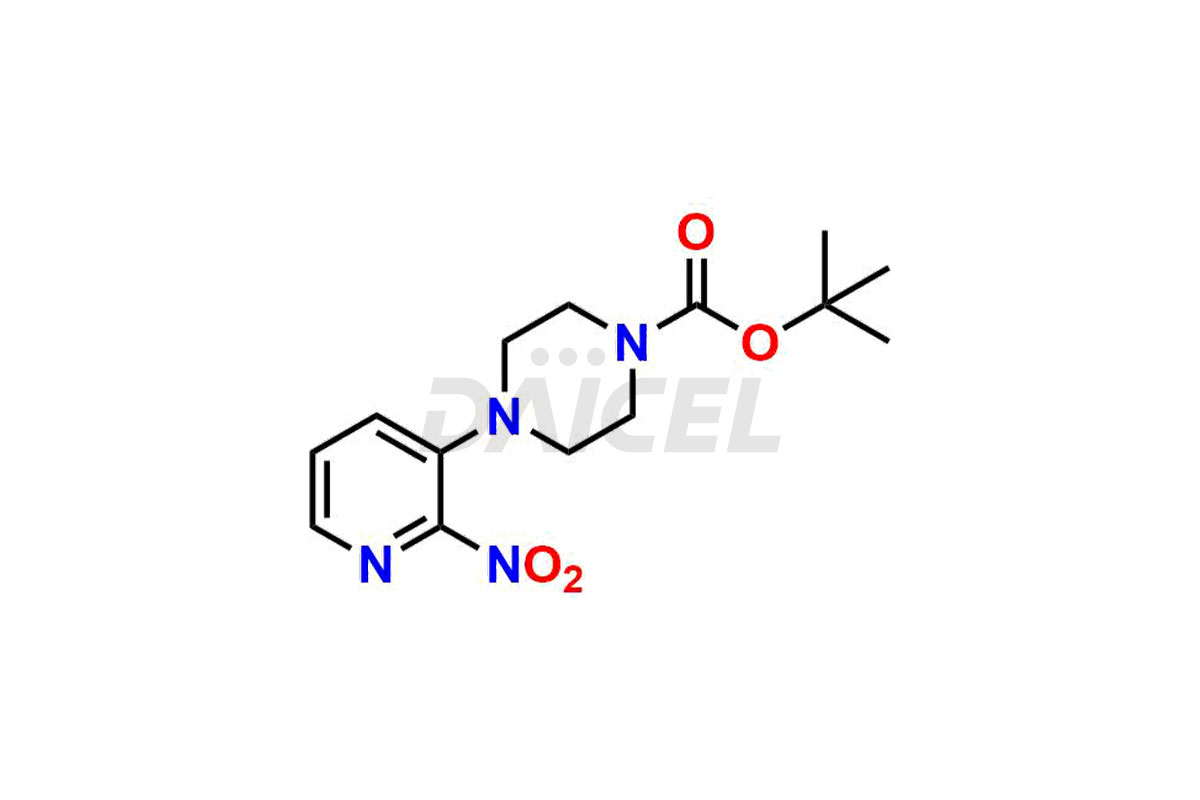
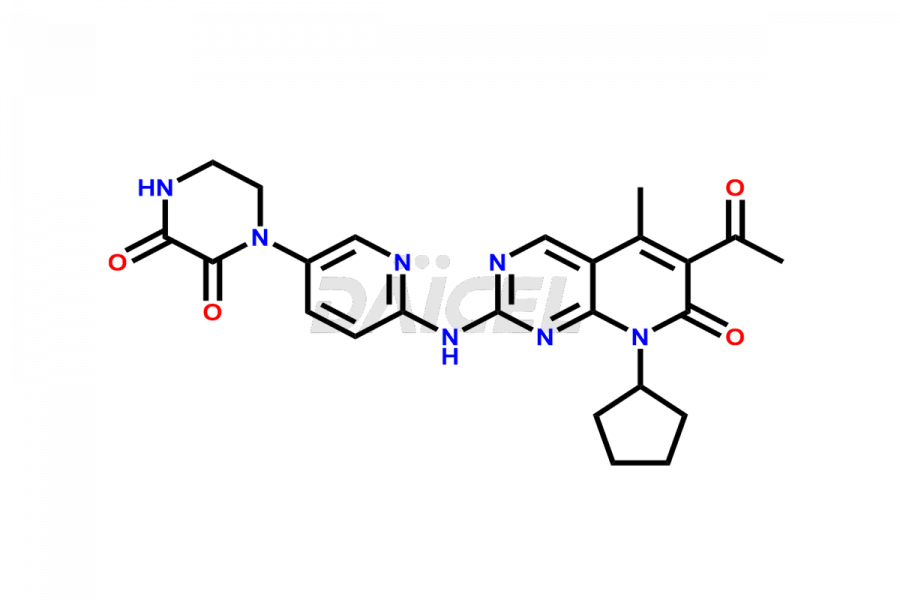
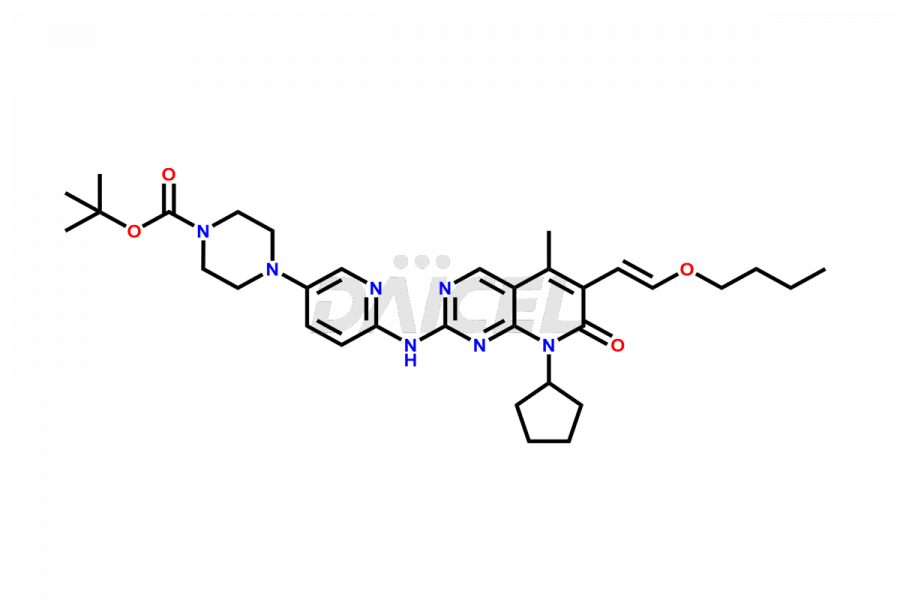
Daicel Pharma synthesizes more than thirty high-quality Palbociclib impurities, such as 6-Desacetyl 6-BromoPalbociclib, 6-Desacetyl Palbociclib, Desoxo-Palbociclib, Palbociclib Glycosamine, Palbociclib Chloro Bromo Impurity, Palbociclib N-Boc Impurity, Palbociclib N-Hydroxy Piperazine, Palbociclib Nitroso Impurity, PBC Amine Isomer Impurity, etc., crucial in determining the quality, stability, and biological safety of the active pharmaceutical ingredient Palbociclib. Moreover, Daicel Pharma offers custom synthesis of Palbociclib impurities and delivers them globally.
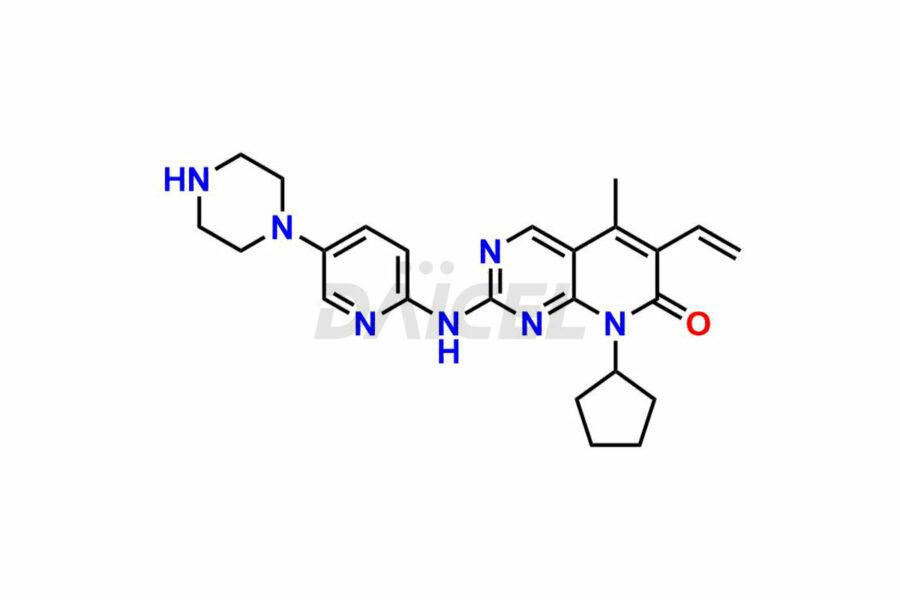
Palbociclib [CAS: 571190-30-2] is a pyrido-pyrimidine derivative, an inhibitor of cyclin-dependent kinases. It is utilized with aromatase inhibitors for treating metastatic breast cancer in postmenopausal women.
Palbociclib: Use and Commercial Availability
Palbociclib is marketed under the trade name Ibrance for treating advanced hormone-receptor-positive breast cancer in women. It is a part of anti-hormonal therapy, either in combination with an aromatase inhibitor or a selective estrogen receptor modulator (SERM). By inhibiting cell division in cancer cells, Palbociclib slows down tumor growth. The drug is available in capsule form in three different doses. 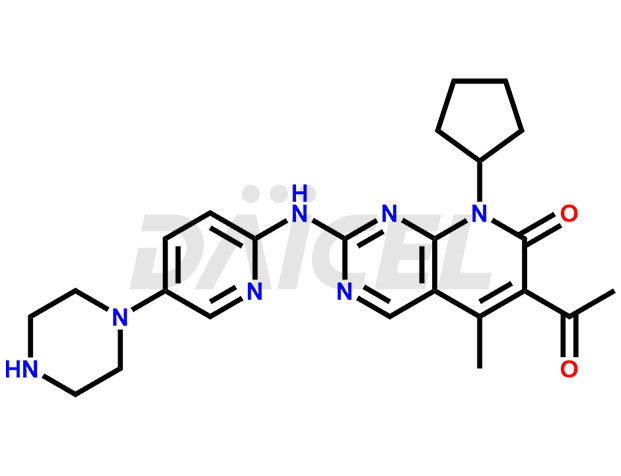
Palbociclib Structure and Mechanism of Action
The chemical name of Palbociclib is 6-Acetyl-8-cyclopentyl-5-methyl-2-[[5-(piperazin-1-yl)pyridin-2-yl]amino]-8H-pyrido[2,3-d]pyrimidin-7-one. The chemical formula for Palbociclib is C24H29N7O2, and its molecular weight is approximately 447.5 g/mol.
Palbociclib inhibits cyclin-dependant kinase (CDK) 4 and 6. This inhibition of CDK4/6 and cyclin D1 kinase activity prevents DNA synthesis and thus inhibits cell division.
Palbociclib Impurities and Synthesis
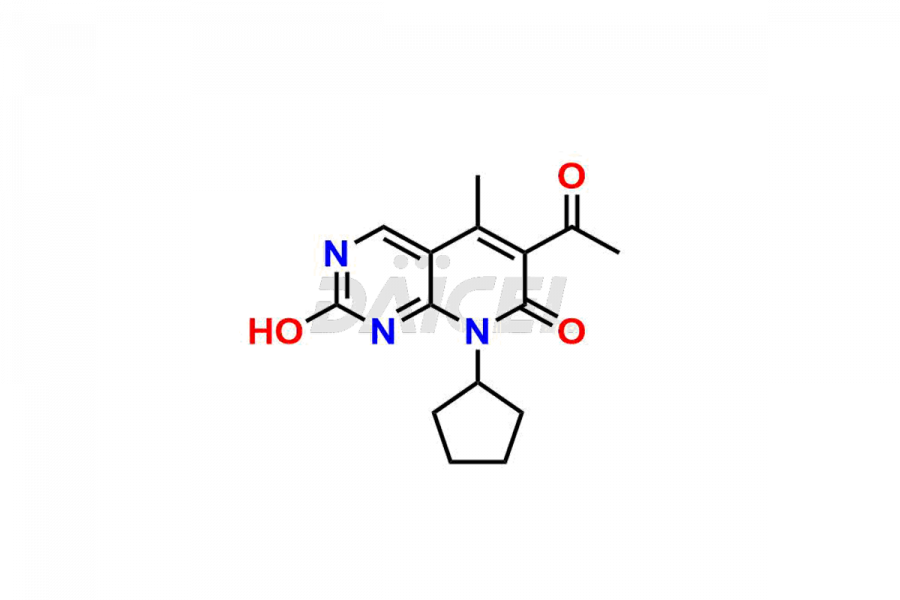
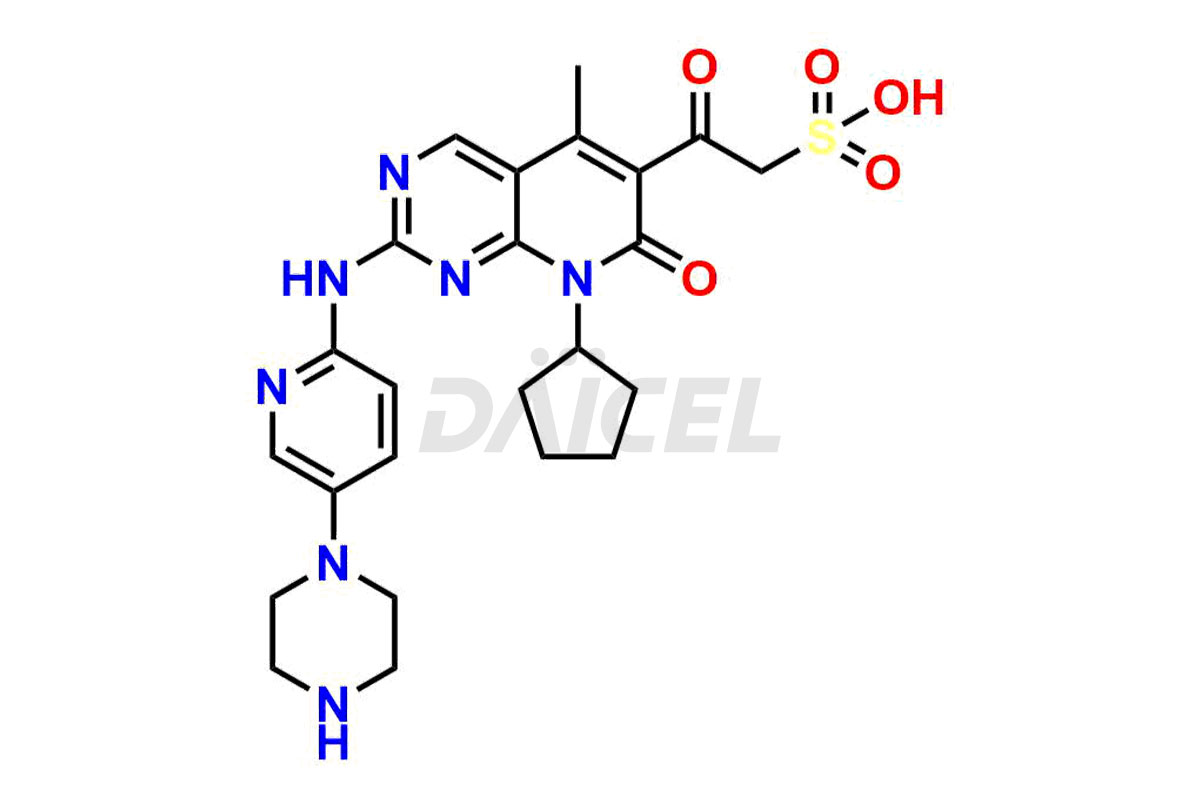
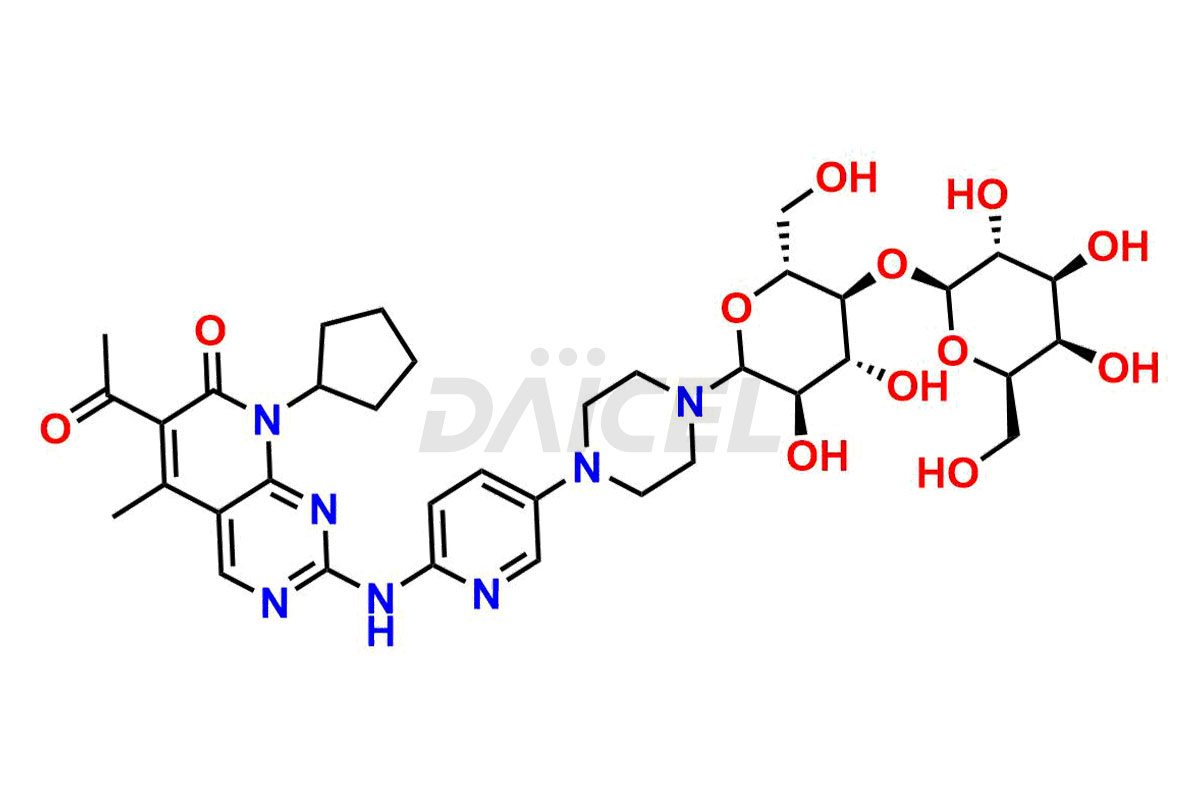
Palbociclib impurities are classified as isomers, degradants, and solvents. Isomers are due to the preparation of stereoisomers in the Palbociclib synthesis process, while degradants result from the instability of Palbociclib caused by hydrolysis, oxidation, or photolysis. Solvents like acetic acid, ethyl acetate, and dimethyl sulfoxide (DMSO) may produce impurities during the synthetic process. For instance, N-desmethyl Palbociclib is an isomeric impurity, while 4-hydroxypiperidin-1-yl Palbociclib is a degradant impurity. These impurities can affect the safety, quality, and efficacy of the final product. Therefore, it is crucial to control impurities during the Palbociclib synthetic1 and purification processes and analyze the final product to ensure it is safe and effective.
Daicel offers a Certificate of Analysis (CoA) from a cGMP-compliant analytical facility for more than thirty Palbociclib impurity standards2 including 6-Desacetyl 6-BromoPalbociclib, 6-Desacetyl Palbociclib, Desoxo-Palbociclib, Palbociclib Glycosamine, Palbociclib Chloro Bromo Impurity, Palbociclib N-Boc Impurity, Palbociclib N-Hydroxy Piperazine, Palbociclib Nitroso Impurity, PBC Amine Isomer Impurity, and more. The CoA includes complete characterization data, such as 1H NMR, 13C NMR, IR, MASS, and HPLC purity. We also provide 13C-DEPT and CHN on request. We also give a complete characterization report on delivery.
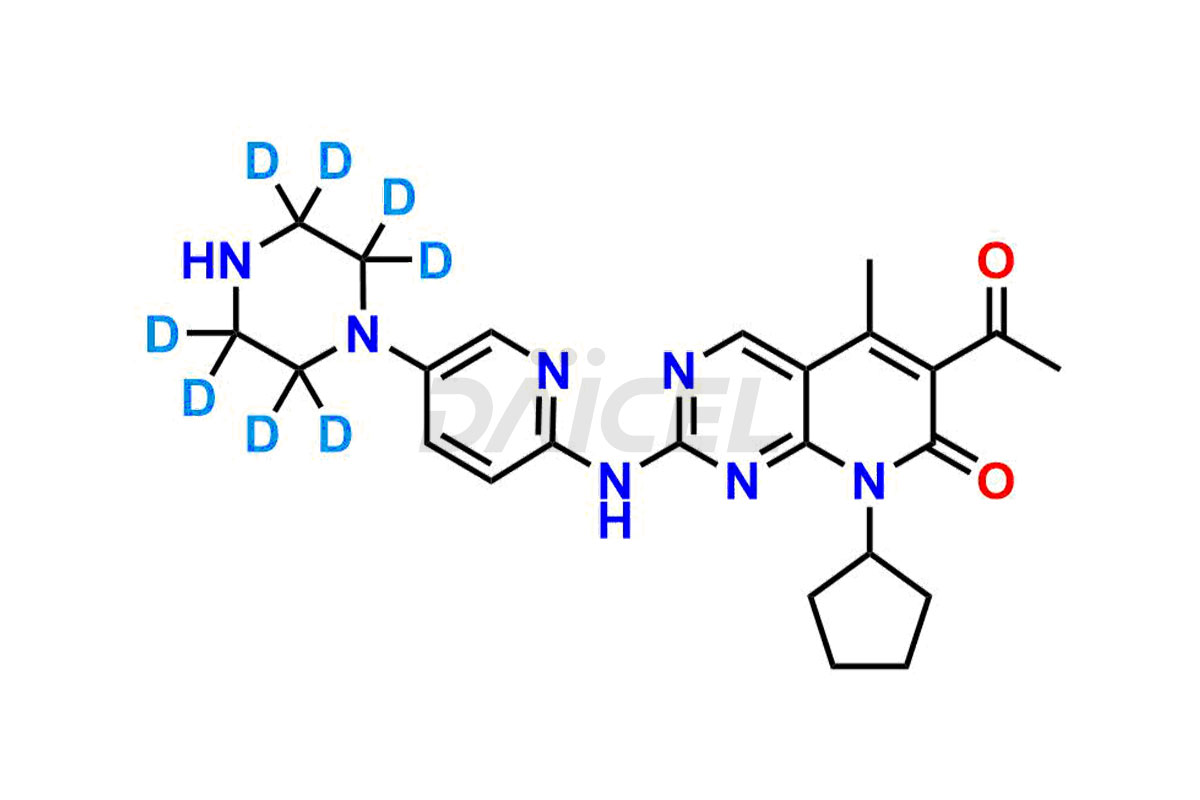
Daicel has the technology and expertise to prepare any unknown Palbociclib impurity or degradation product. The company also provides labeled compounds to quantify the efficacy of generic Palbociclib. Daicel offers highly pure Palbociclib-d8, a deuterium-labeled standard of Palbociclib for bioanalytical research and BA/BE studies with the percentage of isotope data in CoA.