General Information
Norethindrone Impurities and Norethindrone
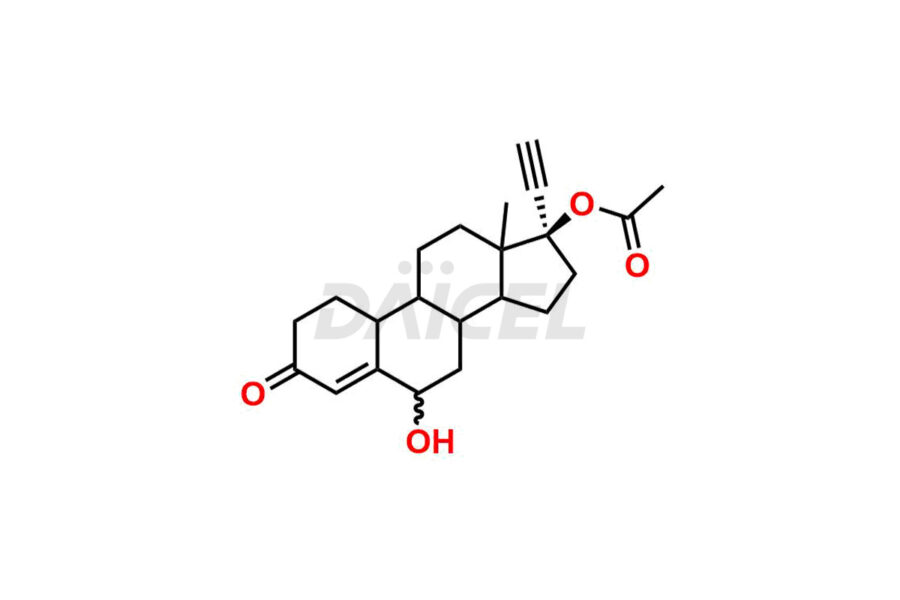
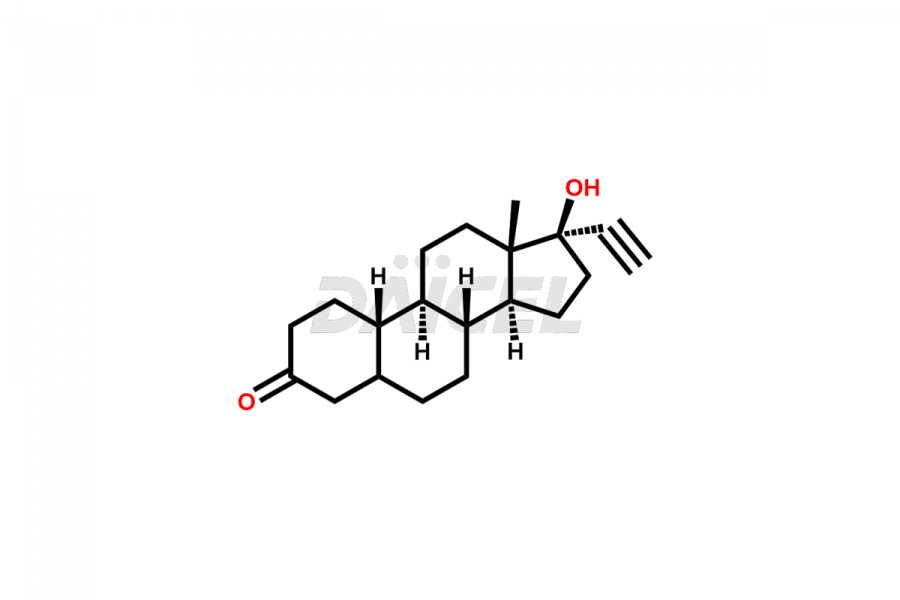
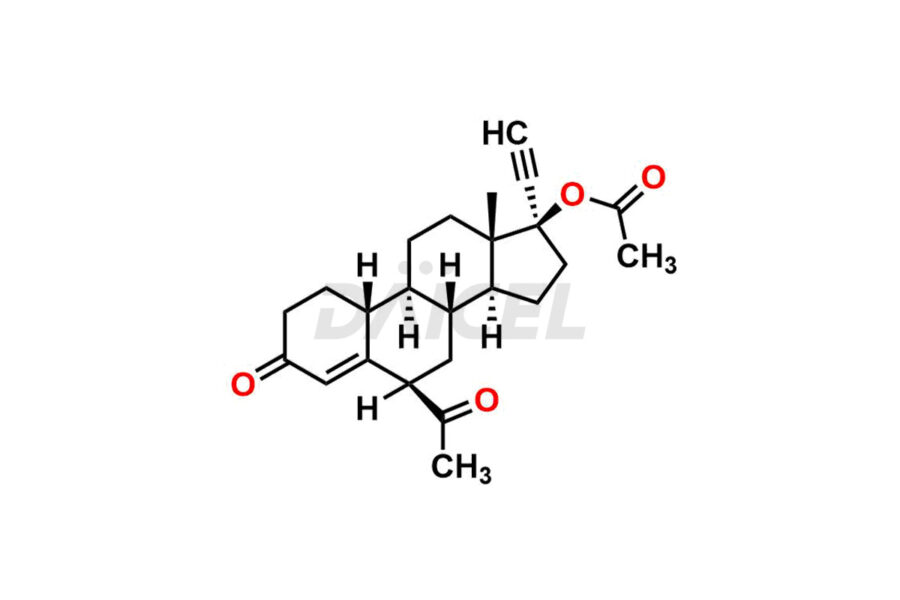
Daicel Pharma offers Norethindrone impurities like 6-Hydroxy Norethindrone Acetate, Dihydro Norethindrone (Mixture of Isomers), and Norethindrone Acetate Impurity-D. These impurities are essential for evaluating the quality, stability, and safety of Norethindrone, which is an active pharmaceutical ingredient. Daicel Pharma provides custom synthesis of Norethindrone impurities and distributes them internationally to fulfill clients’ needs.
Norethindrone [CAS: 68-22-4] is a synthetic progestin used for contraception, treating amenorrhea (absence of menstrual periods), and management of endometriosis.
Norethindrone: Use and Commercial Availability
Norethisterone is approved as an oral contraceptive when used alone or with an estrogen component. It helps in hormone replacement therapy in postmenopausal osteoporosis and vasomotor symptoms associated with menopause. Additionally, it is suitable for treating secondary amenorrhea, endometriosis, and hormonal imbalance-related abnormal uterine bleeding. Norethisterone is available under Camila, Emzahh, Errin, Incassia, Micronor, etc.
Norethindrone Structure and Mechanism of Action 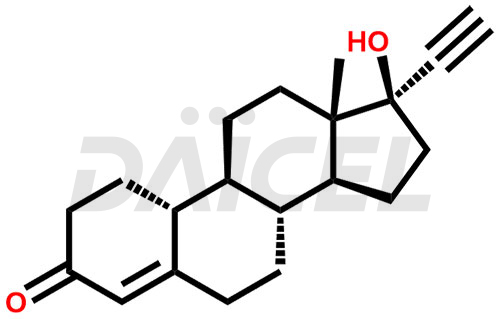
The chemical name of Norethindrone is (17α)-17-Hydroxy-19-norpregn-4-en-20-yn-3-one. Its chemical formula is C20H26O2, and its molecular weight is approximately 298.4 g/mol.
Norethindrone alters cervical mucosa so that it inhibits sperm migration into the uterus. It prevents conception by suppressing ovulation.
Norethindrone Impurities and Synthesis
Norethindrone may contain impurity standards like 6-Hydroxy Norethindrone Acetate, Dihydro Norethindrone (Mixture of Isomers), and Norethindrone Acetate Impurity-D that can arise during the synthesis process or storage. Controlling and monitoring impurities is essential to ensure Norethindrone quality, safety, and efficacy. Stringent quality control measures help meet regulatory standards and ensure the purity of the final product.
Daicel provides a Certificate of Analysis (CoA) of Norethindrone impurity standards. Our cGMP-compliant analytical laboratory offers the CoA, which contains extensive characterization data such as 1H NMR, 13C NMR, IR, MASS, and HPLC purity1,2. We give additional characterization data, such as 13C-DEPT, on request. At Daicel Pharma, we can generate unknown impurities of Norethindrone or its degradation products.