N-Nitroso Lumefantrine Impurity
- CAT NUMBER DCTI-C-2362
- CAS NUMBER NA
- MOLECULAR FORMULA C26H23Cl3N2O2
- MOLECULAR WEIGHT 501.83
Daicel Pharma offers the best-quality Lumefantrine impurities, such as N-Nitroso Lumefantrine Impurity. They are vital for evaluating Lumefantrine quality, stability, and biological safety. Furthermore, Daicel Pharma specializes in the custom synthesis of Lumefantrine impurities and ensures their worldwide delivery.
Lumefantrine [CAS: 82186-77-4] or Benflumetol, is an antimalarial drug. It is a synthetic amino alcohol fluorene derivative. It treats malaria caused by Plasmodium falciparum species. It blocks nucleic acid and protein synthesis by forming a hemin complex.
Lumefantrine treats uncomplicated malaria tropica. It treats patients with chloroquine-resistant malaria. It combines with artemether in treating multi-drug-resistant malaria, especially Plasmodium species. It is a part of the artemisinin-based combination therapy. It is available as an oral formulation under the brand Coartem.
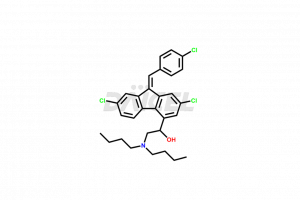
The chemical name of Lumefantrine is (9Z)-2,7-Dichloro-9-[(4-chlorophenyl)methylene]-α-[(dibutylamino)methyl]-9H-fluorene-4-methanol. The chemical formula for Lumefantrine is C30H32cl3NO, and its molecular weight is approximately 528.94 g/mol.
The precise mechanism of action of Lumefantrine is unknown.
During Lumefantrine synthesis, impurities form that may affect drug safety and efficacy. They form during the manufacturing, purification, or storage of Lumefantrine. Therefore, Lumefantrine impurities must be controlled and monitored throughout the drug’s development.
Daicel Pharma offers a comprehensive Certificate of Analysis (CoA) for Lumefantrine impurities, which includes N-Nitroso Lumefantrine Impurity. We provide the CoA from a cGMP-compliant analytical facility. It gives complete characterization data1 such as 1H NMR, 13C NMR, IR, MASS, and HPLC purity2. We provide additional analytical data on request. Daicel Pharma can prepare any unidentified Lumefantrine impurity or degradation product. In addition, Daicel Pharma offers highly purified, stable isotope-labeled standards of Lumefantrine. Clients receive a complete characterization report upon delivery.
The HPLC-DAD method helps to identify and separate Lumefantrine impurities.
Unreacted starting materials, by-products, reagents, and catalysts during synthesis and poor storage conditions may form Lumefantrine impurities and degradation products.
Lumefantrine impurities' presence in the drug can affect its safety, quality, and efficacy.
Note: Products protected by valid patents by a manufacturer are not offered for sale in countries having patent protection. The sale of such products constitutes a patent infringement, and its liability is at the buyer's risk.