General Information
Losartan Impurities and Losartan
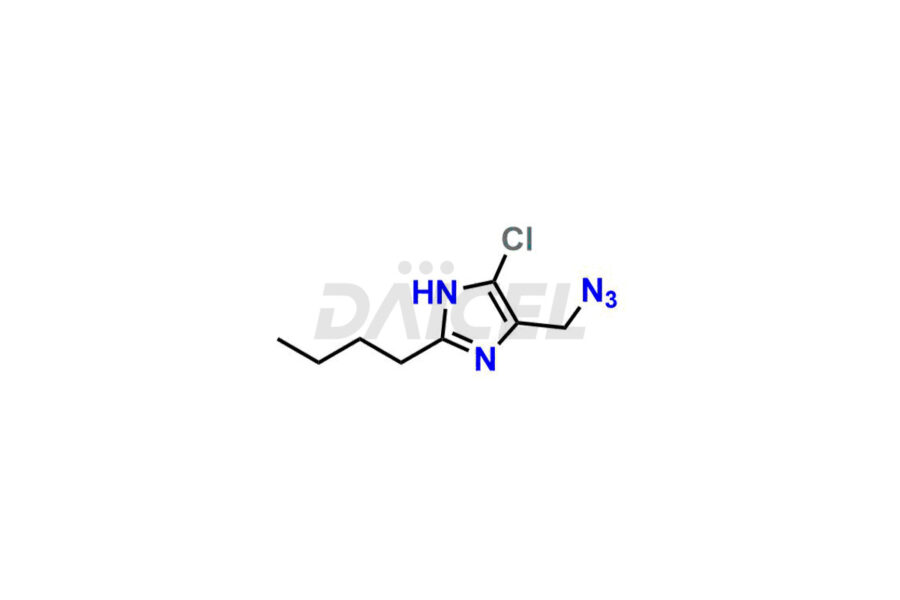
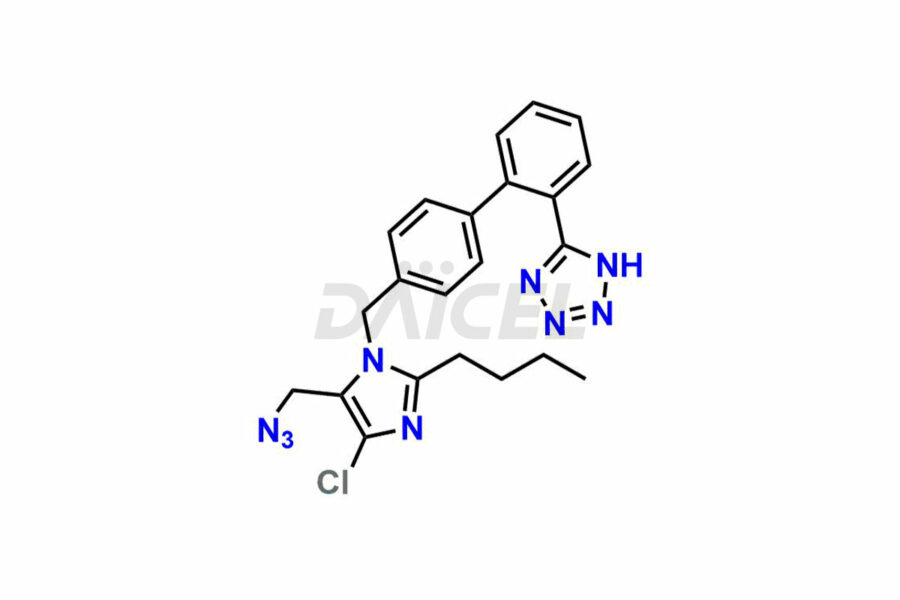
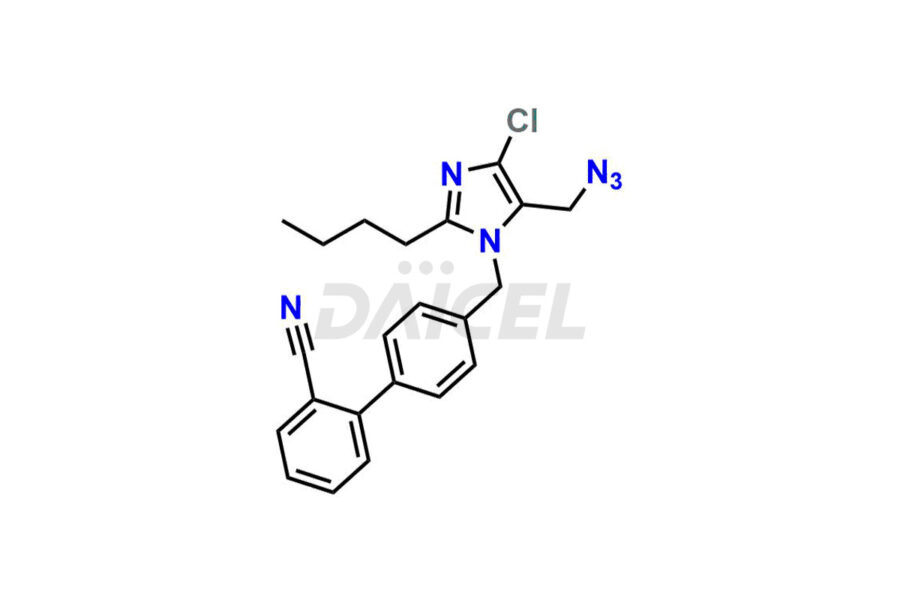
For evaluating the purity and safety of Losartan, an essential active pharmaceutical ingredient, Daicel Pharma offers a customized synthesis of Losartan impurity standards. These impurity standards include crucial compounds such as 4-(azidomethyl)-2-butyl-5-chloro-1H-imidazole, Losartan azide, Losartan Azide Nitrile, and more. Additionally, Daicel Pharma provides worldwide delivery options for Losartan impurity standards.
Losartan [CAS: 114798-26-4], an effective antihypertensive medication, acts as a non-peptide antagonist of the angiotensin II receptor type 1. It treats hypertension and diabetic nephropathy.
Losartan: Use and Commercial Availability
Losartan, marketed under the brand name, Cozaar, is a US FDA-approved medication for various medical conditions. It treats hypertension and diabetic nephropathy. Losartan is an angiotensin II receptor blocker (ARB), and it has renoprotective effects in individuals with type 2 diabetes. Moreover, in cases of hypertension with left ventricular hypertrophy, Losartan inhibits angiotensin II-induced cardiac remodeling and reduces the risk of stroke in these patients.
Losartan Structure and Mechanism of Action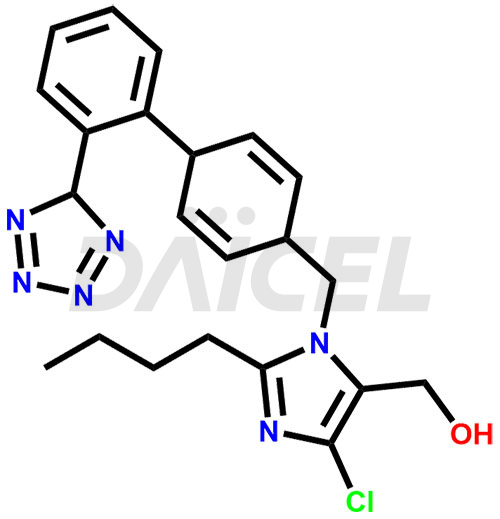
The chemical name of Losartan is 2-Butyl-4-chloro-1-[[2′-(2H-tetrazol-5-yl)[1,1′-biphenyl]-4-yl]methyl]-1H-imidazole-5-methanol. Its chemical formula is C22H23ClN6O, and its molecular weight is approximately 422.9 g/mol.
Losartan is a vasoconstrictor that blocks the binding of angiotensin II to the AT1 receptor found in many tissues.
Losartan Impurities and Synthesis
The analysis and control of impurities in Losartan1, an angiotensin II receptor blocker, is crucial to ensure the quality and safety of the medication. Various Losartan impurities include related substances and degradation products that require identification and quantification. Analytical techniques such as high-performance liquid chromatography (HPLC) and liquid chromatography (LC) analyze these impurities. Stringent control measures and specifications limit the impurity levels in Losartan, ensuring compliance with regulatory requirements.
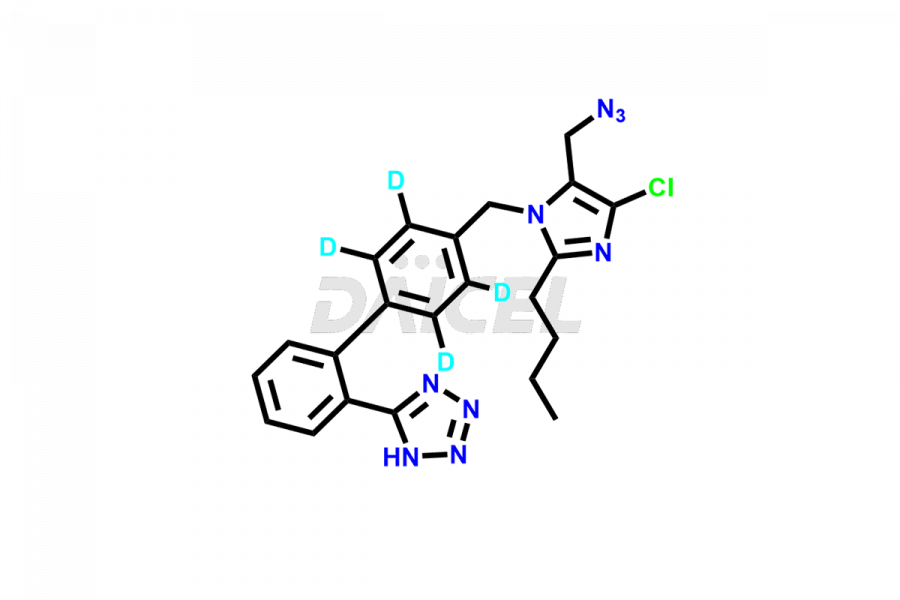
Daicel Pharma strictly adheres to cGMP standards and operates an analytical facility for the preparation of Losartan impurity standards, which include 4-(azidomethyl)-2-butyl-5-chloro-1H-imidazole, Losartan azide, Losartan Azide Nitrile, and so on. In addition, we offer deuterium-labeled Losartan compound, Losartan Impurity 21-D4, which is essential for conducting bioanalytical research and BA/BE studies. Our Losartan impurity standards have a detailed Certificate of Analysis (CoA) that provides a comprehensive characterization report. This report includes data obtained through techniques, 1H NMR, 13C NMR, IR, MASS, and HPLC purity analysis2. Upon request, we give additional data like 13C-DEPT. Moreover, we can synthesize unknown Losartan impurity standards, degradation products, and labeled compounds to evaluate the effectiveness of generic Losartan. Each delivery has a comprehensive characterization report.