General Information
Lopinavir Impurities and Lopinavir
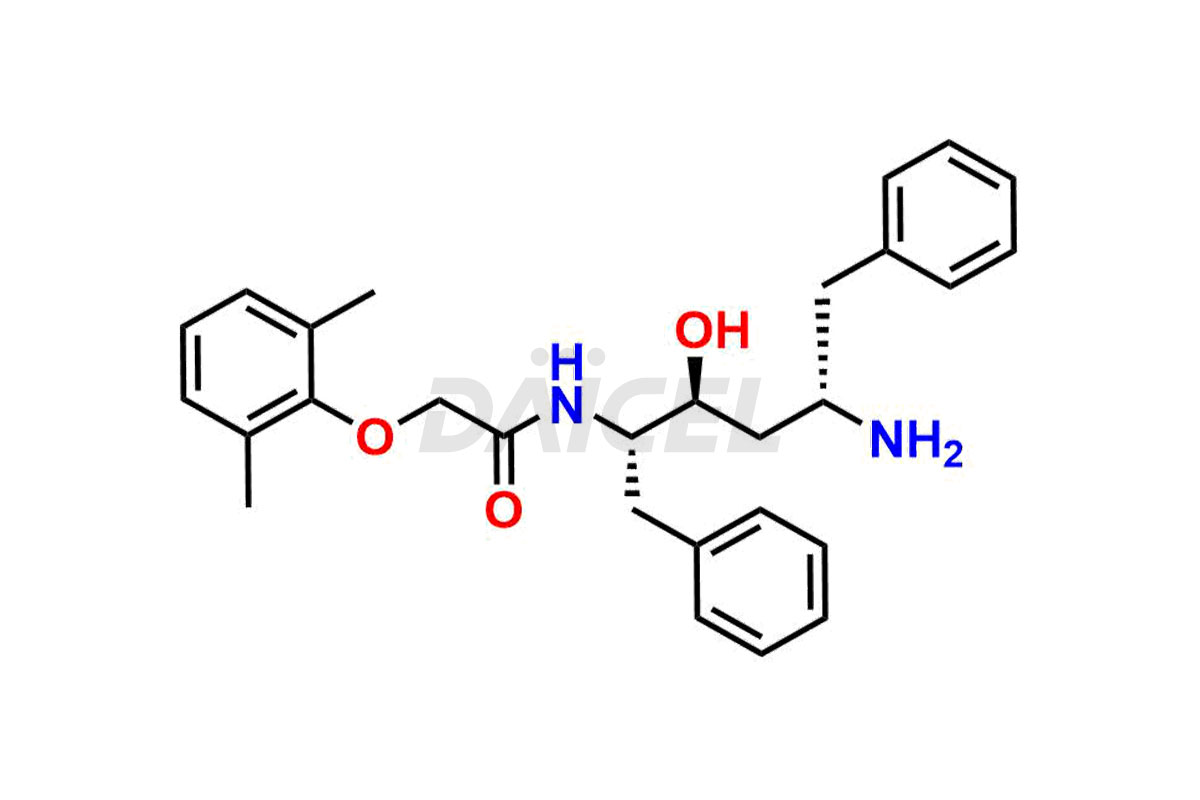
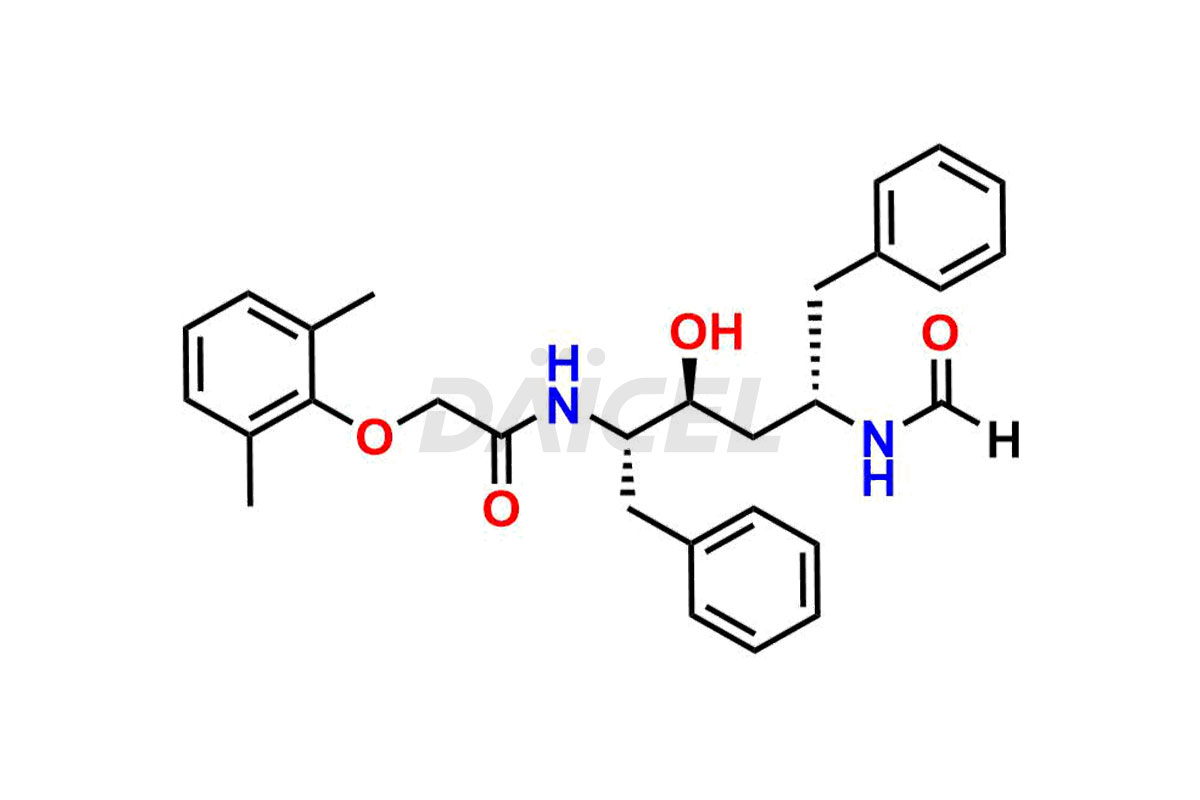
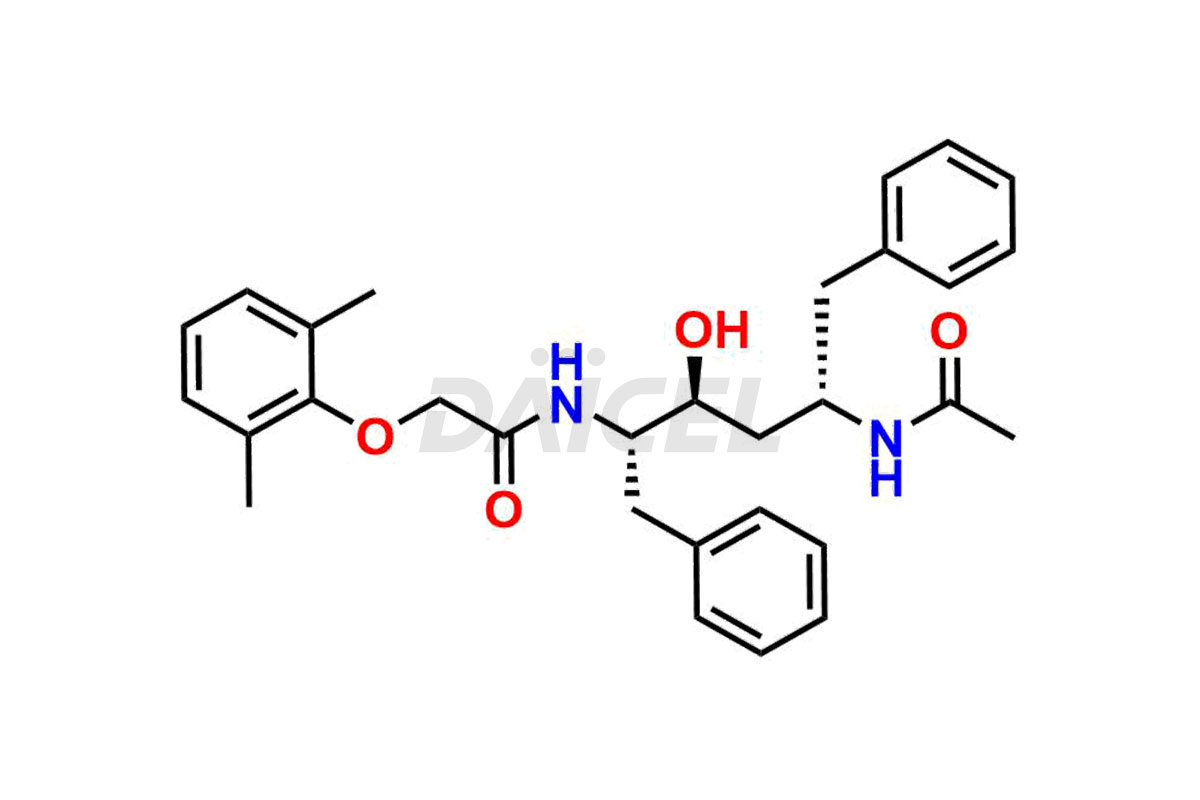
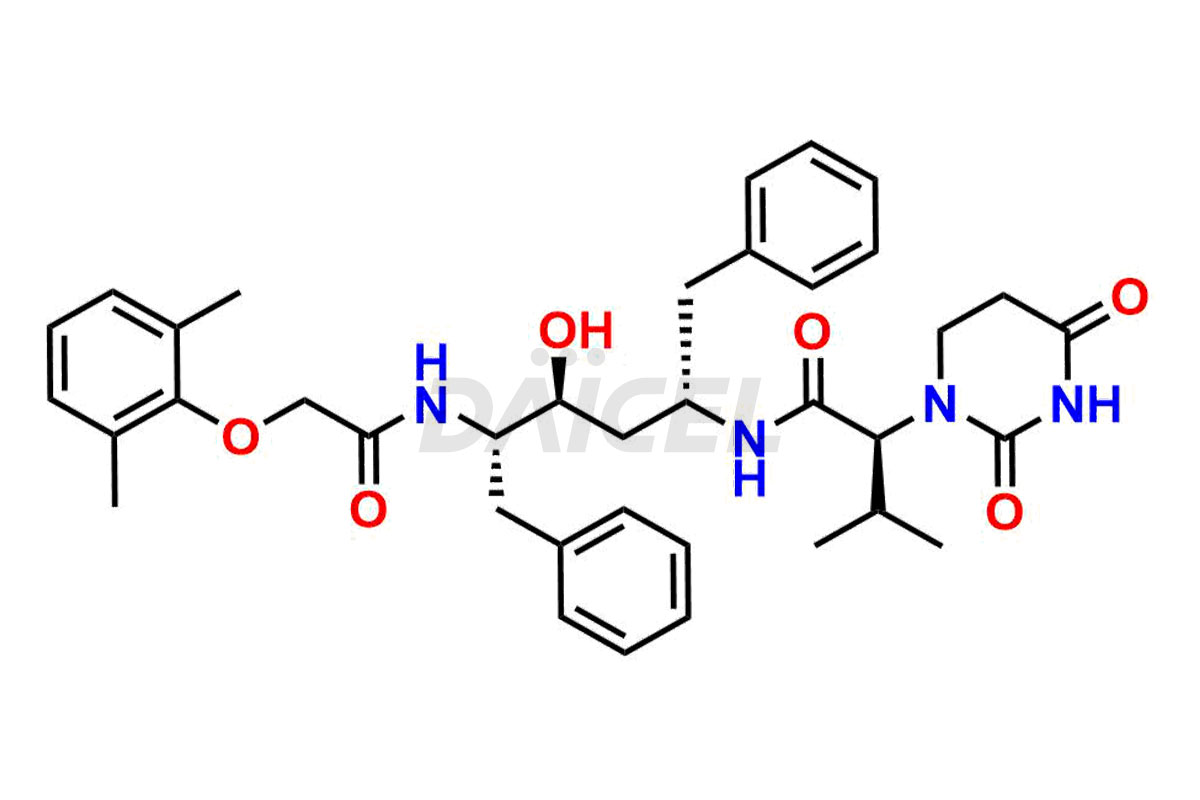
For evaluating the purity and safety of Lopinavir, an essential active pharmaceutical ingredient, Daicel Pharma offers a customized synthesis of Lopinavir impurity standards. These impurity standards include crucial compounds such as Lopinavir EP Impurity-E, Lopinavir EP Impurity-F, Lopinavir EP Impurity-G, Lopinavir Metabolite M1 Impurity, and Lopinavir Metabolite M3-M4 Impurity. Additionally, Daicel Pharma provides worldwide delivery options for Lopinavir impurity standards.
Lopinavir [CAS: 192725-17-0] is an antiretroviral protease inhibitor in combination with ritonavir for treating and preventing HIV infection and AIDS. It is a peptidomimetic HIV protease inhibitor that remains effective against HIV protease even in the presence of the Val 82 mutation.
Lopinavir: Use and Commercial Availability
Kaletra is the brand name for the combination product Lopinavir/Ritonavir, which combines with antiretrovirals and treats HIV-1 infection in adults and pediatric patients ≥14 days old. This product consists of an HIV protease inhibitor, Lopinavir, and the inhibitor of cytochrome P-450 CYP3A, Ritonavir.
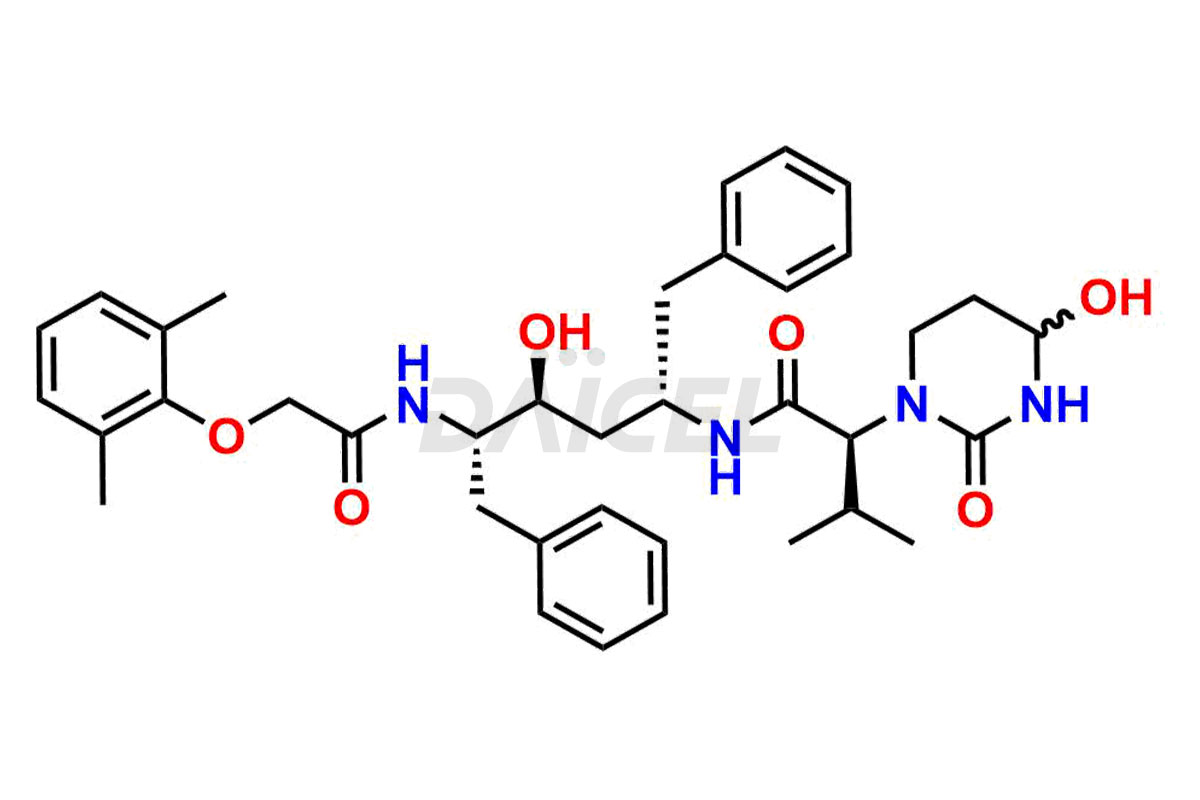
Lopinavir Structure and Mechanism of Action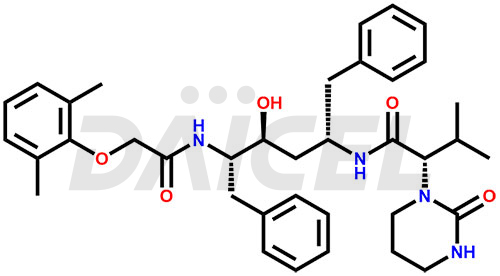
The chemical name of Lopinavir is (αS)-N-[(1S,3S,4S)-4-[[2-(2,6-Dimethylphenoxy)acetyl]amino]-3-hydroxy-5-phenyl-1-(phenylmethyl)pentyl]tetrahydro-α-(1-methylethyl)-2-oxo-1(2H)-pyrimidineacetamide. Its chemical formula is C37H48N4O5, and its molecular weight is approximately 628.8 g/mol.
Lopinavir prevents the cleavage of the Gag-Pol polyprotein. It inhibits the preparation of immature, non-infectious viral particles.
Lopinavir Impurities and Synthesis
Impurities in Lopinavir are unintended substances that can be present in the drug product with the active pharmaceutical ingredient (API). They may originate from manufacturing1, starting materials, or interactions with environmental factors. Common Lopinavir impurities include related compounds, degradation products, residual solvents, and process-related impurities. It is crucial to monitor and control the levels of these impurities during manufacturing to ensure the safety, efficacy, and quality of the Lopinavir drug product. Regulatory authorities have established guidelines and limits for impurity levels to maintain purity and minimize potential risks associated with the drug. Stringent quality control measures minimize impurity levels and ensure product integrity.
Daicel Pharma strictly adheres to cGMP standards and operates an analytical facility for preparing Lopinavir impurity standards, which include Lopinavir EP Impurity-E, Lopinavir EP Impurity-F, Lopinavir EP Impurity-G, Lopinavir Metabolite M1 Impurity, and Lopinavir Metabolite M3-M4 Impurity. Our Lopinavir impurity standards have a detailed Certificate of Analysis (CoA) that provides a comprehensive characterization report. This report includes data obtained through techniques, 1H NMR, 13C NMR, IR, MASS, and HPLC purity analysis2. Upon request, we give additional data like 13C-DEPT. Moreover, we can synthesize unknown Lopinavir impurity standards and degradation products. Each delivery has a comprehensive characterization report.