6,7-DIMETHOXYISATIN
- CAT NUMBER DCTI-C-2891
- CAS NUMBER 76159-91-6
- MOLECULAR FORMULA C10H9NO4
- MOLECULAR WEIGHT 207.19
Daicel Pharma offers excellent-quality Isoproterenol impurities, such as 6,7-DIMETHOXYISATIN. Isoproterenol impurities are critical for evaluating the quality, stability, and biological safety. In addition, Daicel Pharma is a specialist in preparing Isoproterenol impurities and ensures worldwide delivery.
Isoproterenol [CAS: 7683-59-2] or Isoprenaline is a brochodilator. It is an analog of epinephrine that dilates the bronchi and increases the heart rate. It is a beta-1 and beta-2 adrenergic receptor agonist, which relaxes the smooth muscles of the bronchi, uterus, and Gastrointestinal tract. Further, it is a trace-amine-associated receptor 1 (TAAR1) agonist.
Isoproterenol treats patients with heart block, not requiring pacemakers. Further, it is for cardiac arrest cases when pacemakers are unavailable. It helps patients with congestive heart failure, septic shock, bronchospasm during anesthesia, bradycardia, etc. Isoproterenol increases the cardiac output without vasoconstriction. Its administration is through the intravenous route. Isoproterenol is available under various brands like Aerolone, Isuprel, Norisodrine, Vapo-iso, etc.
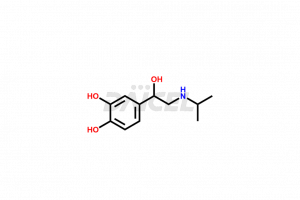
The chemical name of Isoproterenol is 4-[1-Hydroxy-2-[(1-methylethyl)amino]ethyl]-1,2-benzenediol. The chemical formula for Isoproterenol is C11H17NO3, and its molecular weight is approximately 211.26 g/mol.
As a beta-adrenergic receptor agonist, Isoproterenol activates G-protein coupled receptors. It leads to an increase in intracellular cAMP and activates protein kinase A (PKA). PKA phosphorylates cardiac L-type calcium channels that depolarize cells by inward active movement of calcium ions.
During Isoproterenol synthesis1, impurities form that may affect the safety and efficacy of the drug. They form during the synthetic process, purification, or storage of Isoproterenol. Therefore, Isoproterenol impurities must be controlled and monitored throughout the drug development.
Daicel Pharma offers a comprehensive Certificate of Analysis (CoA) for Isoproterenol impurities, which includes 6,7-DIMETHOXYISATIN. We provide the CoA from a cGMP-compliant analytical facility. It gives complete characterization data2 such as 1H NMR, 13C NMR, IR, MASS, and HPLC purity. We provide additional analytical data on request. Daicel Pharma can prepare any unidentified Isoproterenol impurity or degradation product. In addition, Daicel Pharma offers highly purified, stable isotope-labeled standards of Isoproterenol. The clients of Daicel Pharma can expect a complete characterization report on delivery.
HPLC, LC/ESI-MS, and NMR analytical methods help identify the process-related Isoproterenol impurities.
Oxidation processes, light exposure, and nucleophilic reactions cause degradation of Isoproterenol.
Secondary amines and nitrates lead to the formation of nitrosamine impurities in Isoproterenol.
Note: Products protected by valid patents by a manufacturer are not offered for sale in countries having patent protection. The sale of such products constitutes a patent infringement, and its liability is at the buyer's risk.