Glycopyrrolate EP Impurity L
- CAT Number DCTI-C-1697
- CAS Number 19833-96-6
- Molecular Formula C14H18O3
- Molecular Weight 234.3
Daicel Pharma offers superior-quality Glycopyrrolate impurities, such as Glycopyrrolate EP Impurity L. These impurities are essential for evaluating Glycopyrrolate quality, stability, and biological safety. In addition, Daicel Pharma specializes in the custom synthesis of Glycopyrrolate impurities and ensures their worldwide delivery.
Glycopyrrolate [CAS: 596-51-0] or Glycopyrronium, inhibits acetylcholine receptors. It is a quaternary ammonium compound with antispasmodic properties. It is a pre-operative medicine that stops salivary and respiratory secretions. It blocks acetylcholine effects in the central nervous system, smooth muscles, and secretory glands.
As an anticholinergic agent, Glycopyrrolate reduces salivary and pharyngeal secretion in patients. It reduces chronic drooling in pediatric patients with neurological conditions like cerebral palsy. It treats primary axillary hyperhidrosis in patients. In addition, it prevents and treats peptic ulcers. Many companies manufacture Glycopyrrolate in different formulations. It is available under the brands Robinul, Seebri, Cuvposa, etc.
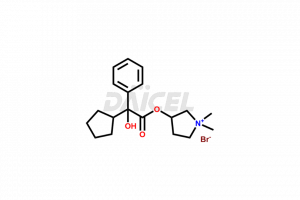
The chemical name of Glycopyrrolate is 3-[(2-cyclopentyl-2-hydroxy-2-phenylacetyl)oxy]-1,1-dimethyl- Pyrrolidinium bromide. The chemical formula for Glycopyrrolate is C19H28NO3.Br and its molecular weight is approximately 398.33 g/mol.
Glycopyrrolate prevents acetylcholine action on salivary glands, thus reducing the rate of salivation. Further, it inhibits acetylcholine’s effects in tissues at the parasympathetic levels.
While synthesizing Glycopyrrolate 1, impurities may form that will affect drug safety and efficacy. They form during the synthetic process, storage, or purification of Glycopyrrolate. Manufacturers can control and monitor Glycopyrrolate impurities to improve the drug’s safety, efficacy, and storage.
Daicel Pharma offers a comprehensive Certificate of Analysis (CoA) for Glycopyrrolate impurities, which includes Glycopyrrolate EP Impurity L. The CoA provided to clients is from a cGMP-compliant analytical facility and has the complete characterization data2 such as 1H NMR, 13C NMR, IR, MASS, and HPLC purity. We give additional spectral data on request. Daicel Pharma can prepare any unidentified Glycopyrrolate impurity or degradation product. In addition, Daicel Pharma offers highly purified, stable isotope-labeled standards of Glycopyrrolate. Daicel Pharma provides a complete characterization report accompanying the delivery.
An ion-pair HPLC and RP-HPLC methods help to analyze Glycopyrrolate impurities.
Glycopyrrolate degrades during storage and on exposure to heat and light.
Glycopyrrolate impurities may affect the drug quality, safety, and efficacy. Hence, it is crucial to remove them.
Note: Products protected by valid patents by a manufacturer are not offered for sale in countries having patent protection. The sale of such products constitutes a patent infringement, and its liability is at the buyer's risk.