General Information
Etomidate Impurities and Etomidate
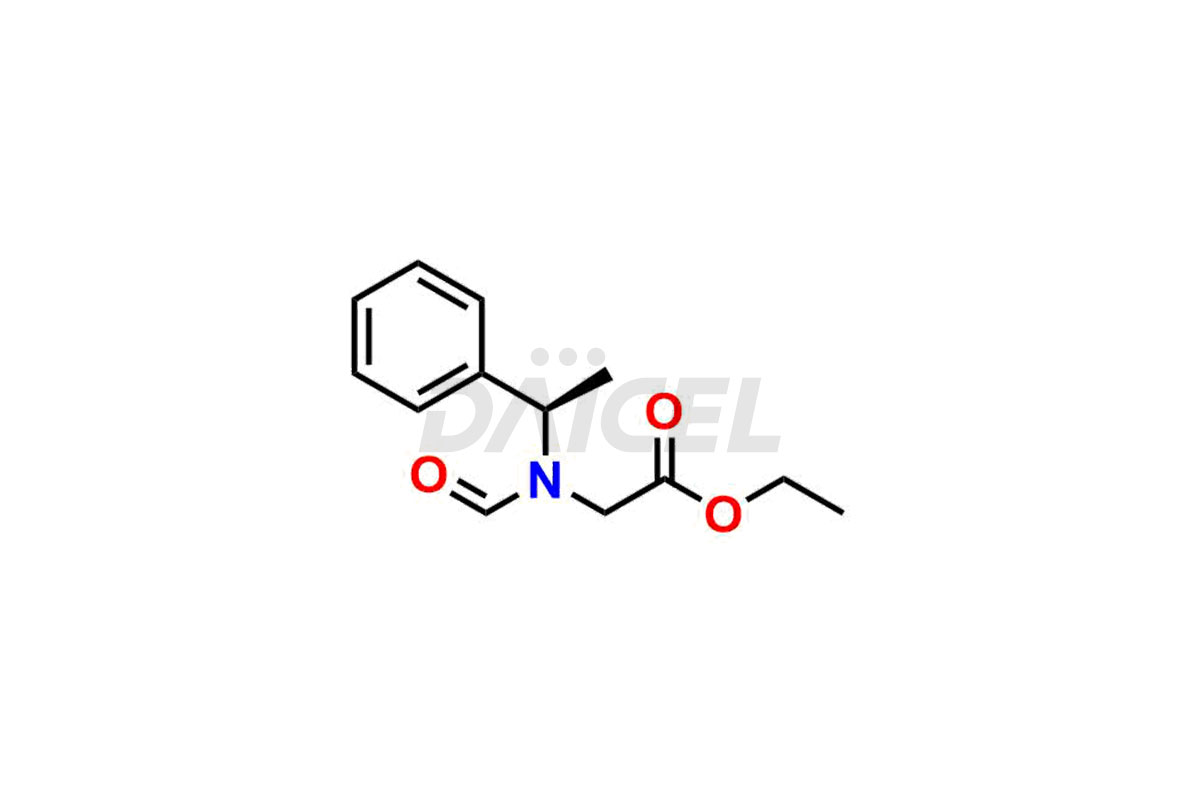
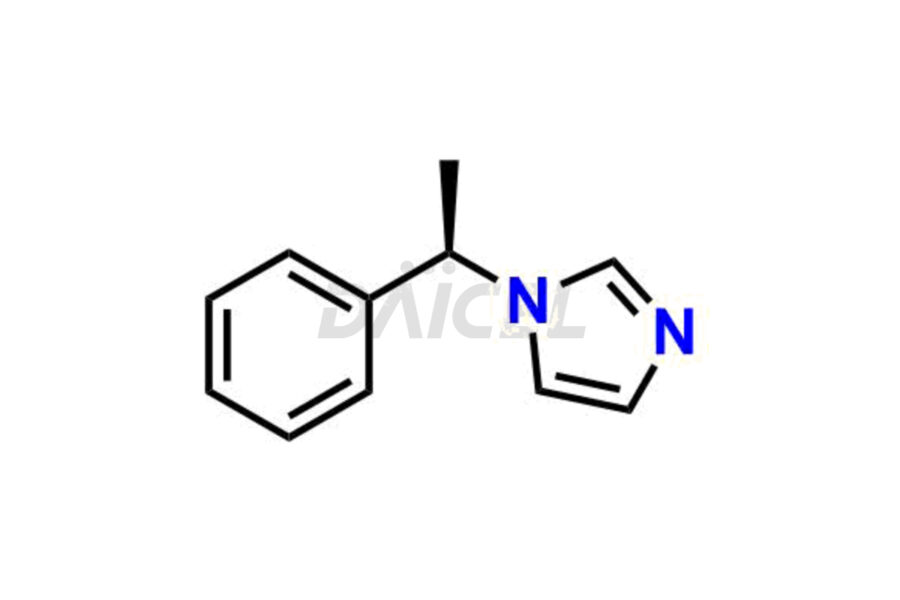
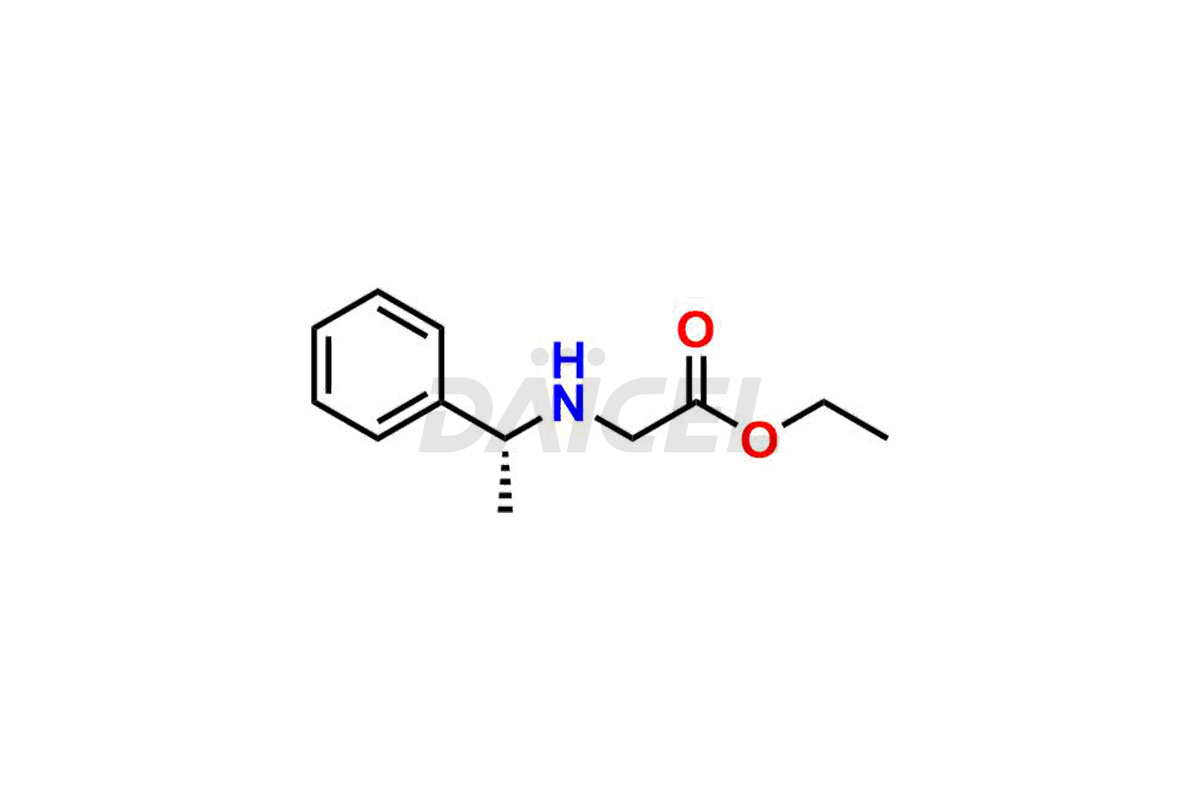
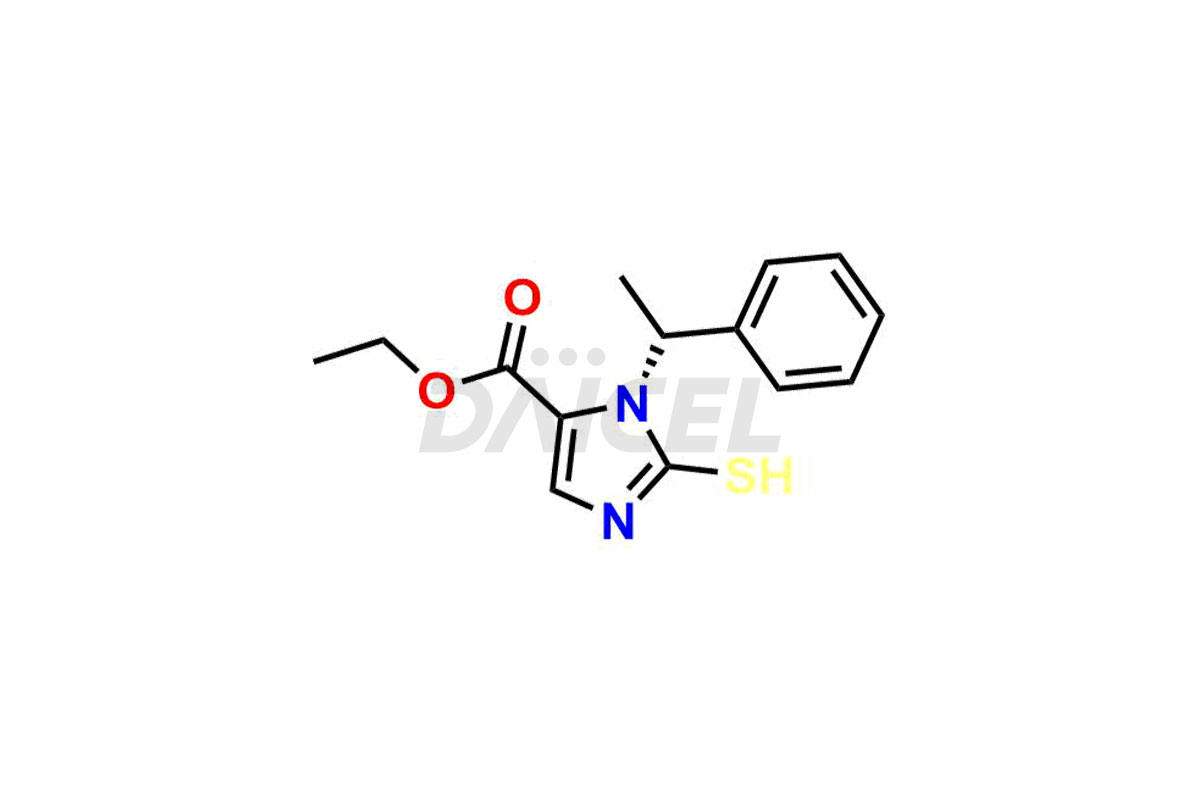
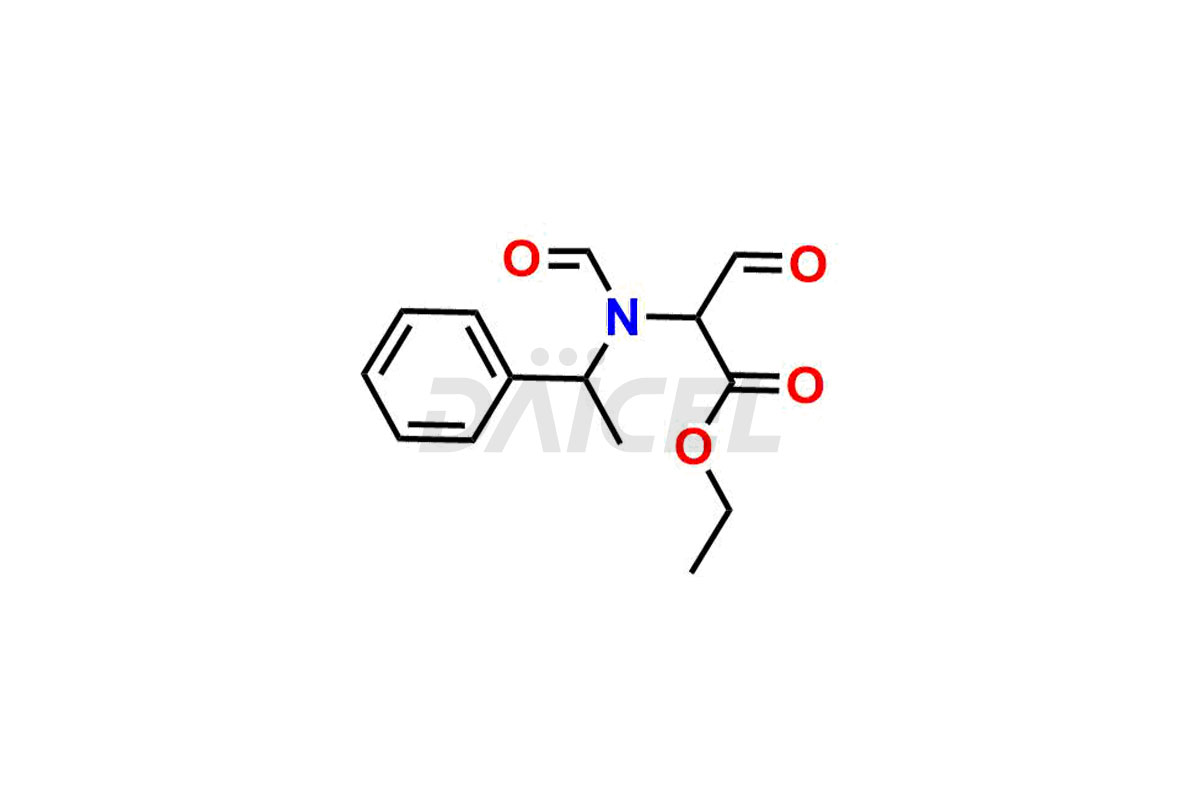
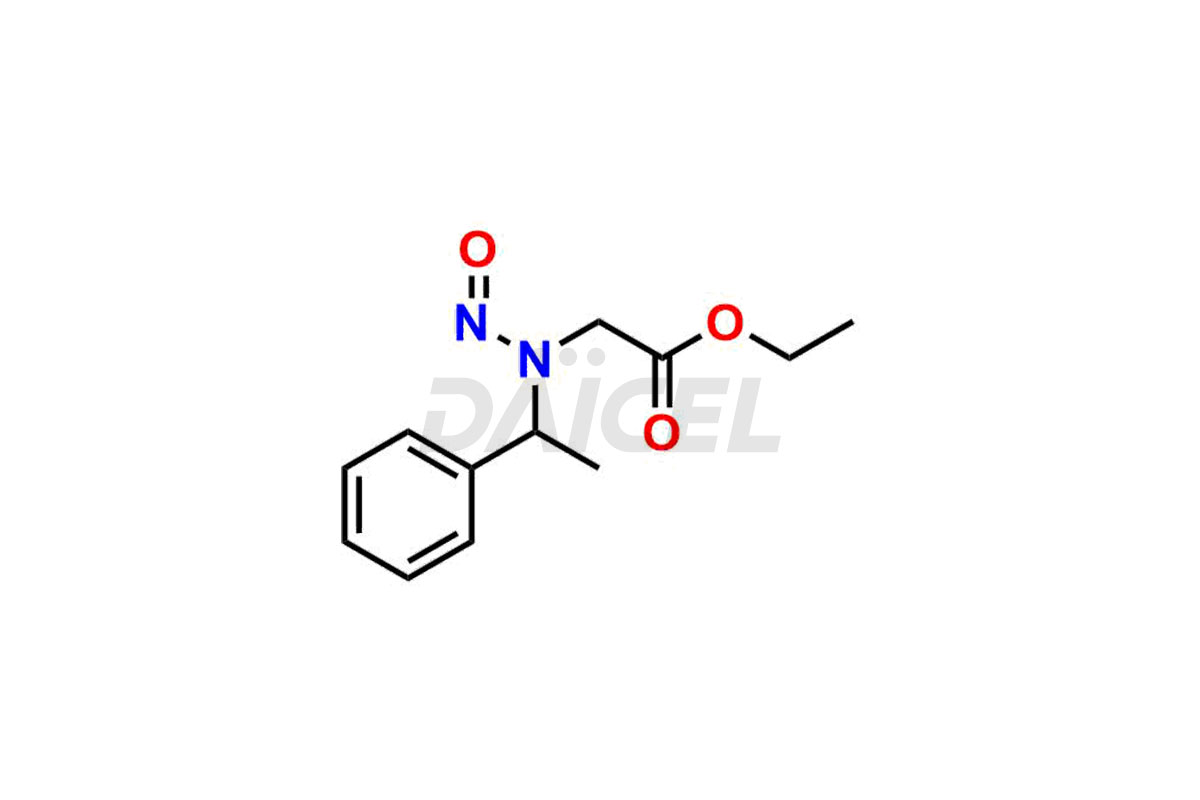
Daicel Pharma synthesizes high-quality Etomidate impurities, (R)-ethyl N-formyl-N-(1-phenylethyl) glycine, 1H-imidazole, 1-[(1R)-1-phenylethyl]-, Ethyl (R)-[(1-Phenylethyl)amino] acetate, Ethyl N-formyl-3-oxo-N-(1-phenylethyl)alaninate, Etomidate intermediate Nitroso impurity, and Ethyl (R)-3-(1-phenylethyl)-2-thioxo-23-dihydro-1H-imidazole-4-carboxylate., crucial in analyzing the quality, stability, and biological safety of the active pharmaceutical ingredient, Etomidate. Moreover, Daicel Pharma offers custom synthesis of Etomidate impurities and delivers them globally.
Etomidate [CAS: 33125-97-2] is a short-acting anesthetic. It is an imidazole carboxylate derivative.
Etomidate: Use and Commercial Availability
Etomidate is an anesthetic agent that acts quickly and has no analgesic properties. It is available under the trade name AMIDATE (intravenous administration). It is an ultrashort-acting, non-barbiturate hypnotic drug used during general anesthesia, rapid sequence intubation, and procedural sedation. Etomidate is also used to maintain anesthesia and short surgical procedures, such as reducing dislocated joints, cardioversion, and tracheal intubation.
Etomidate Structure and Mechanism of Action
The chemical name of Etomidate is (R)-Ethyl 1-(1-phenylethyl)-1H-imidazole-5-carboxylate. Its chemical formula is C14H16N2O2, and its molecular weight is approximately 244.29 g/mol.
Etomidate is structurally different from other anesthetic agents. It interacts with gamma-Aminobutyric acid type A (GABAA) receptors, potentiating the effects of GABA.
Etomidate Impurities and Synthesis
Impurities in the drug substance and finished drug product may impact its safety and efficacy. So, the synthesis of Etomidate impurities helps identify, quantify, and evaluate the chemical properties and stability of the drug. Further, developing analytical methods to detect and quantify impurities is critical for drug quality control and regulatory compliance.
Daicel offers a Certificate of Analysis (CoA) from a cGMP-compliant analytical facility for Etomidate impurity standards, (R)-ethyl N-formyl-N-(1-phenylethyl) glycine, 1H-imidazole, 1-[(1R)-1-phenylethyl]-, Ethyl (R)-[(1-Phenylethyl)amino] acetate, Ethyl N-formyl-3-oxo-N-(1-phenylethyl)alaninate, Etomidate intermediate Nitroso impurity, and Ethyl (R)-3-(1-phenylethyl)-2-thioxo-23-dihydro-1H-imidazole-4-carboxylate. The CoA includes complete characterization data, such as 1H NMR, 13C NMR, IR, MASS, and HPLC purity1,2. We also provide 13C-DEPT and CHN on request. We give a complete characterization report on delivery. Daicel has the technology and expertise to prepare any unknown Etomidate impurity or degradation product.