General Information
Emtricitabine Impurities and Emtricitabine
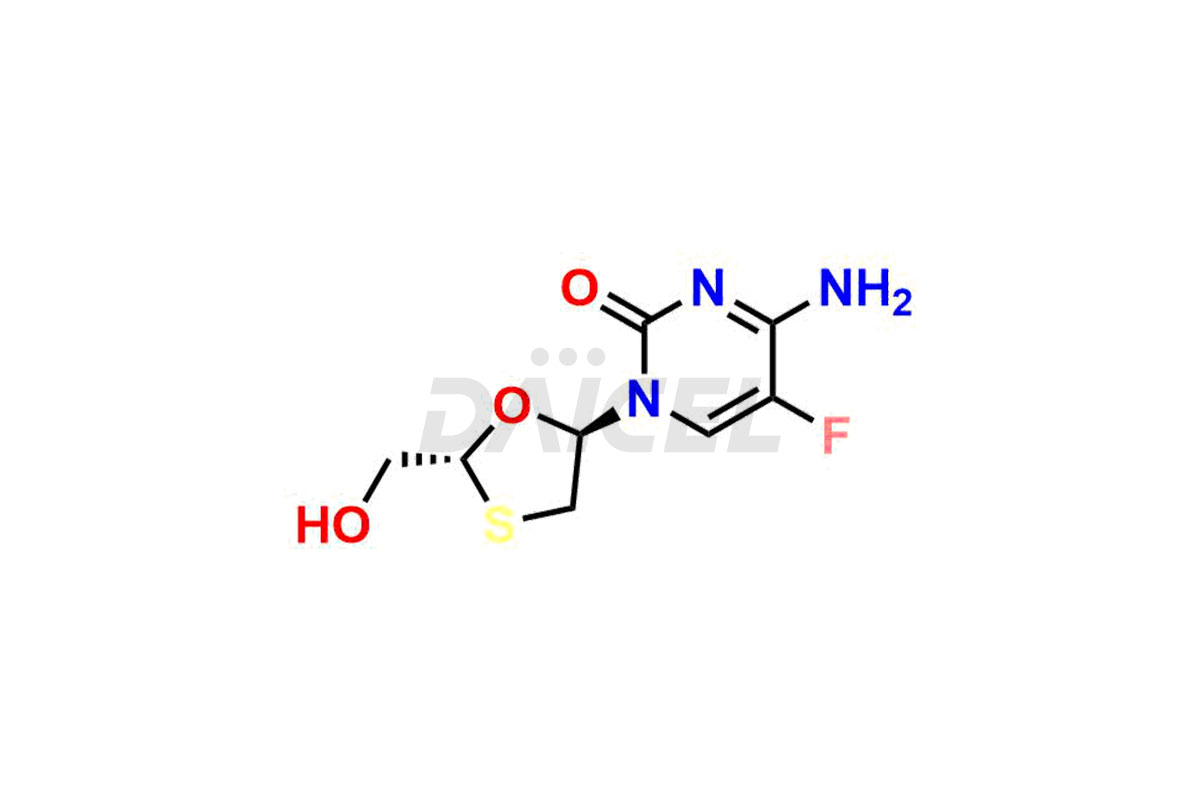
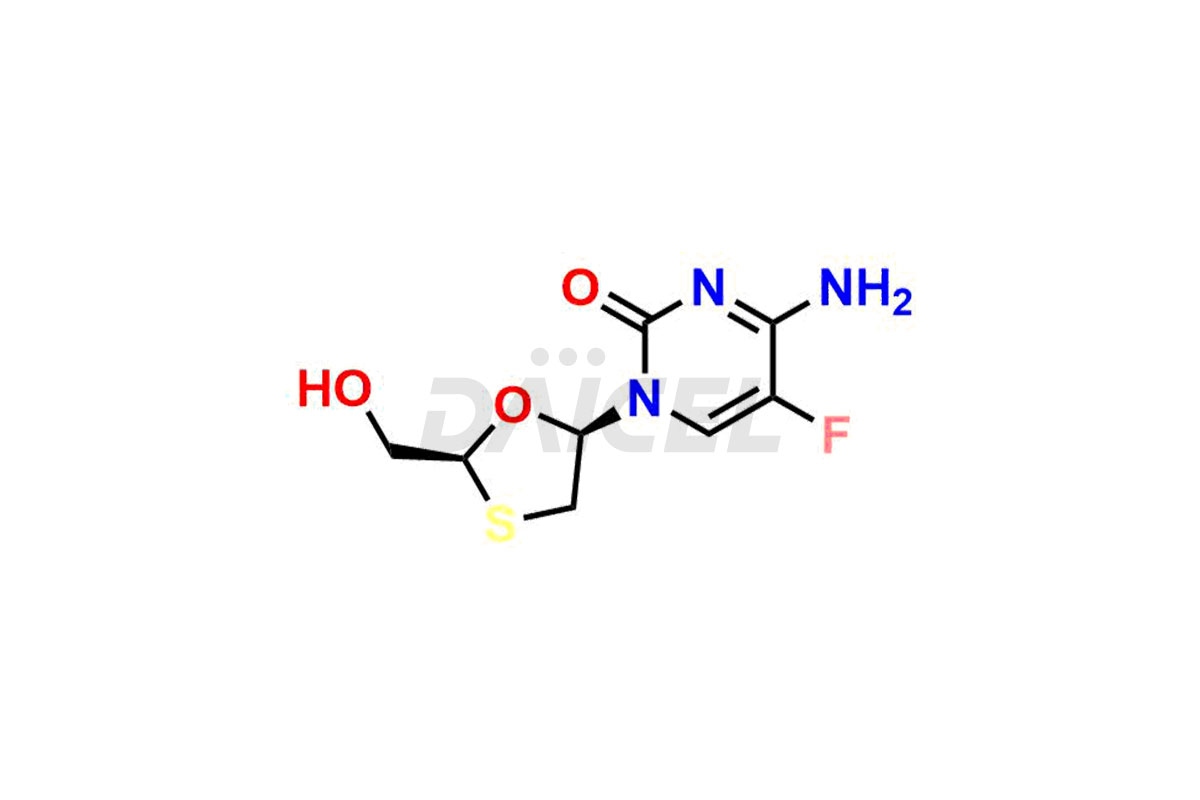
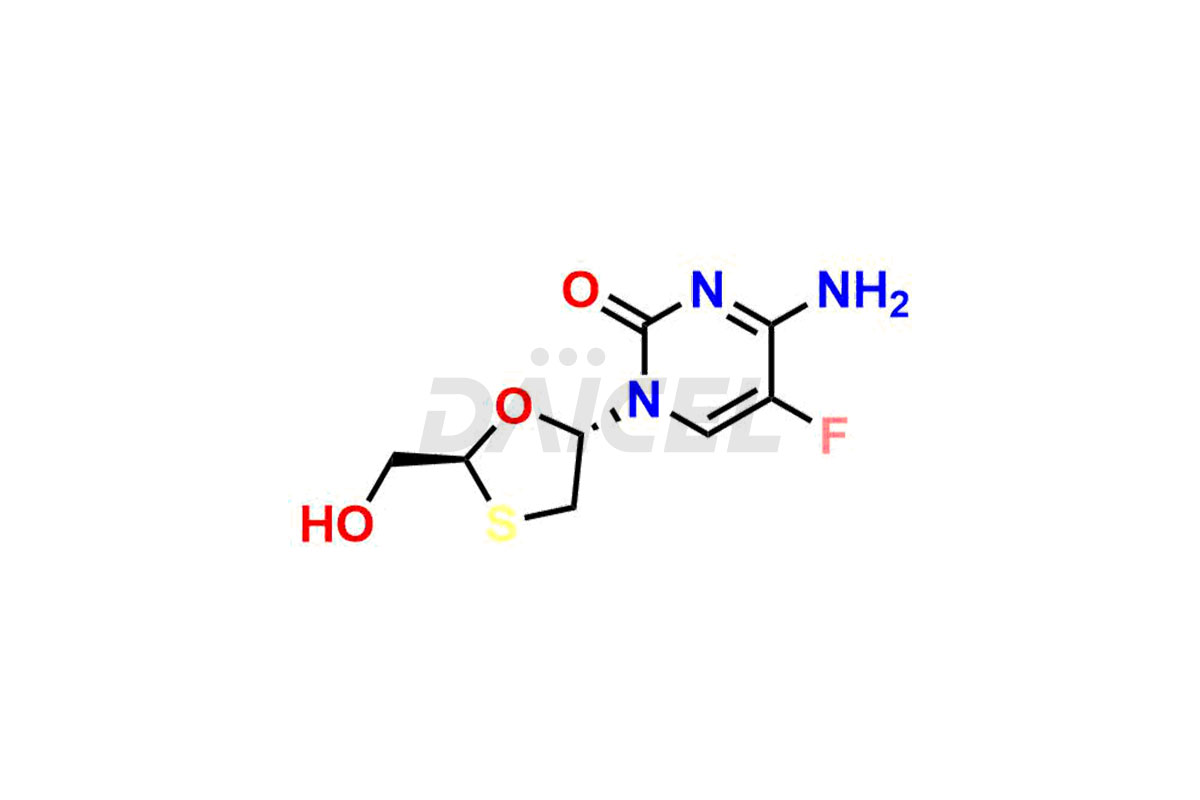
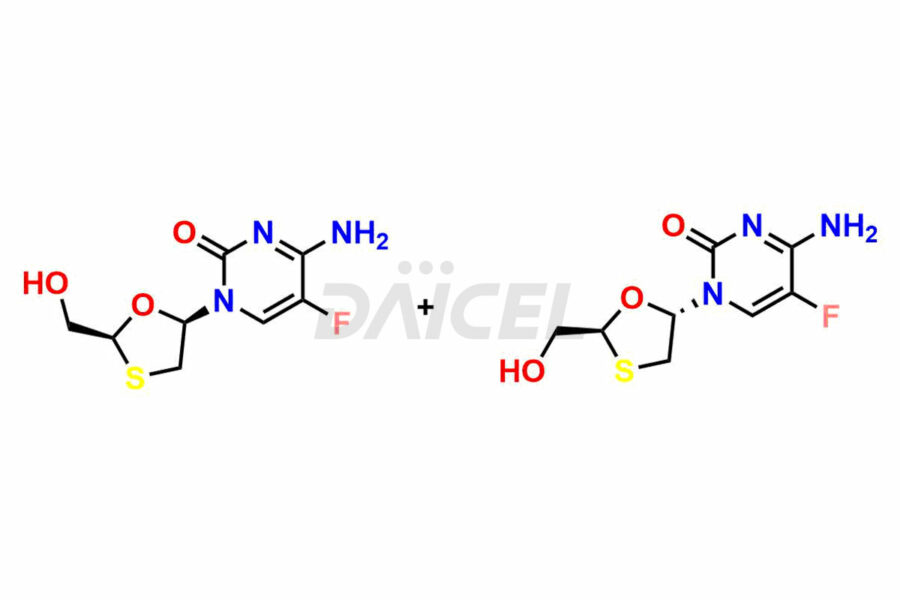
Daicel Pharma synthesizes high-quality Emtricitabine impurities, (2R,5R)-Emtricitabine, (2S,5R)-Emtricitabine, (2S,5S)-Emtricitabine and Emtricitabine Diastereomer, which are crucial in the analysis of the quality, stability, and biological safety of the active pharmaceutical ingredient Emtricitabine. Moreover, Daicel Pharma offers custom synthesis of Emtricitabine impurities and delivers them globally.
Emtricitabine [CAS: 143491-57-0] is a synthetic nucleoside analog possessing antiviral properties. It is the (-) enantiomer of a thio analog of cytidine.
Emtricitabine: Use and Commercial Availability
Emtricitabine is a nucleoside reverse transcriptase inhibitor. It is effective against both human immunodeficiency virus (HIV) and hepatitis B virus. , In combination with other antiretroviral drugs, Emtricitabine treats HIV infection in adults. It effectively reduces or maintains viral load suppression in antiretroviral naïve adults or experienced patients switching from stable combination regimes. Emtricitabine is a preferred choice for patients coinfected with HIV and hepatitis B virus. It is an active ingredient in drugs such as Emtriva, Atripla, Biktarvy, Complera, Descovy Genvoya, Odefsey, Stribild, Symtuza, Truvada, and others.
Emtricitabine Structure and Mechanism of Action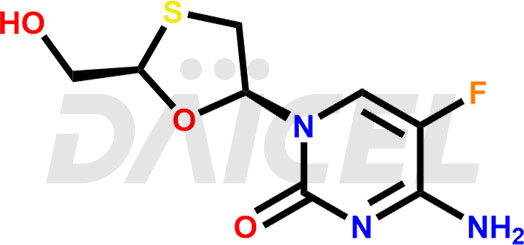
The chemical name of Emtricitabine is 4-Amino-5-fluoro-1-[(2R,5S)-2-(hydroxymethyl)-1,3-oxathiolan-5-yl]-2(1H)-pyrimidinone. Its chemical formula is C8H10FN3O3S, and its molecular weight is approximately 247.25 g/mol.
Emtricitabine is phosphorylated by cellular enzymes giving emtricitabine 5’-triphosphate. It inhibits the activity of HIV-1 reverse transcriptase and incorporates it into nascent viral DNA resulting in chain termination.
Emtricitabine Impurities and Synthesis
During the synthesis1 and subsequent storage of Emtricitabine, impurities can form through various mechanisms such as oxidation, deamination, and isomerization. These impurities may affect the stability and efficacy of Emtricitabine, and their presence must be monitored and controlled during manufacturing.
Daicel offers a Certificate of Analysis (CoA) from a cGMP-compliant analytical facility for Emtricitabine impurity standards, (2R,5R)-Emtricitabine, (2S,5R)-Emtricitabine, (2S,5S)-Emtricitabine, and Emtricitabine Diastereomer. The CoA includes complete characterization data, such as 1H NMR, 13C NMR, IR, MASS, and HPLC purity2. We also provide 13C-DEPT and CHN on request. We also give a complete characterization report on delivery. Daicel has the technology and expertise to prepare any unknown Emtricitabine impurity or degradation product.