General Information
Duloxetine Impurities and Duloxetine
Daicel Pharma offers high-quality impurities for Duloxetine, an active pharmaceutical ingredient. The impurity, Duloxetine Alcohol, plays a vital role in assessing the purity, reliability, and safety of Duloxetine. Daicel Pharma also offers a customized synthesis of Duloxetine impurities to cater to client requirements, with worldwide delivery options available.
Duloxetine [CAS: 116539-59-4] is a selective serotonin and norepinephrine reuptake inhibitor (SNRI). It acts as an antidepressant and for managing neuropathic pain. It is a thiophene derivative. Furthermore, it treats pain in patients with diabetes mellitus and fibromyalgia.
Duloxetine: Use and Commercial Availability
Duloxetine is a medication of the class of serotonin and norepinephrine reuptake inhibitors (SNRIs) and has diverse applications for managing various conditions. It treats major depressive disorder (MDD), generalized anxiety disorder (GAD), chronic musculoskeletal pain, diabetic peripheral neuropathy, and fibromyalgia. The drug is available under brand names such as Cymbalta, Yentreve, Drizalma Sprinkle, Nodetrip, and Ariclaim.
Duloxetine Structure and Mechanism of Action 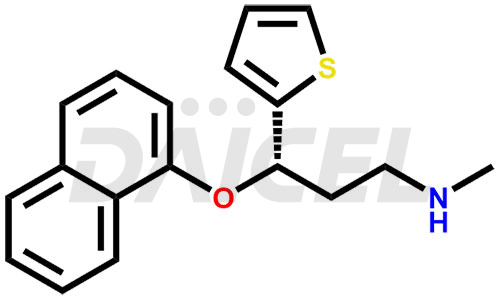
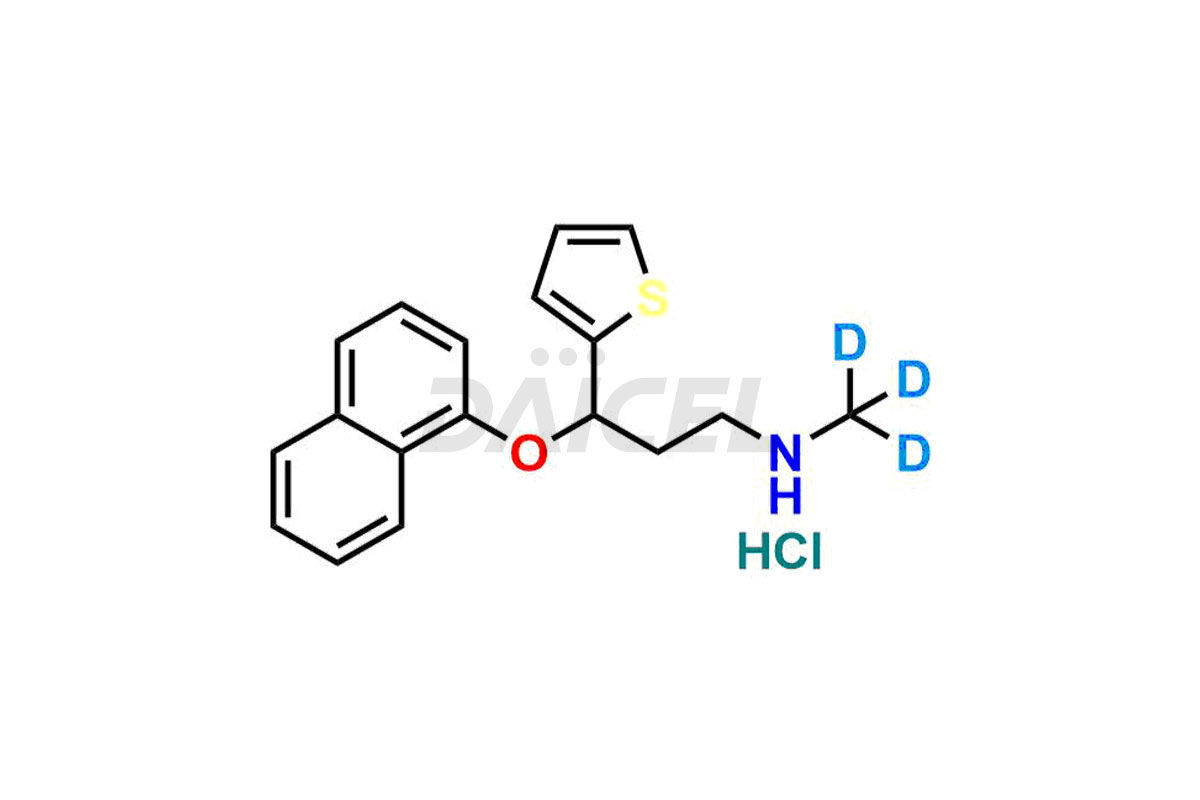
The chemical name of Duloxetine is (S)-(+)-N-Methyl-3-(1-naphthyloxy)-3-(2-thienyl)propylamine. Its chemical formula is C18H19NOS, and its molecular weight is approximately 297.4 g/mol.
Duloxetine inhibits neuronal serotonin and norepinephrine uptake. It has noradrenergic activity in the CNS.
Duloxetine Impurities and Synthesis
Ensuring the quality and safety of Duloxetine drug requires diligent synthesis, analysis, and control of Duloxetine impurities. They can be unintended substances that arise during the manufacturing process1. Rigorous analytical techniques, such as high-performance liquid chromatography (HPLC), are for identifying and quantifying impurities. Strict control measures help minimize the presence of Duloxetine impurities, adhering to regulatory standards and maintaining the purity of the final product.
Daicel Pharma offers a comprehensive Certificate of Analysis (CoA) for the Duloxetine impurity standard, Duloxetine Alcohol. The impurity is from an analytical facility that complies with cGMP standards. The CoA provides a detailed characterization report with data obtained through techniques such as 1H NMR, 13C NMR, IR, MASS, and HPLC purity analysis2. We give additional data like 13C-DEPT upon request. Daicel Pharma synthesizes unknown Duloxetine impurities or degradation products. Duloxetine.HCl-D3, a deuterium-labeled Duloxetine compound, is available for bio-analytical research, including BA/BE studies. Every delivery has a complete characterization report.