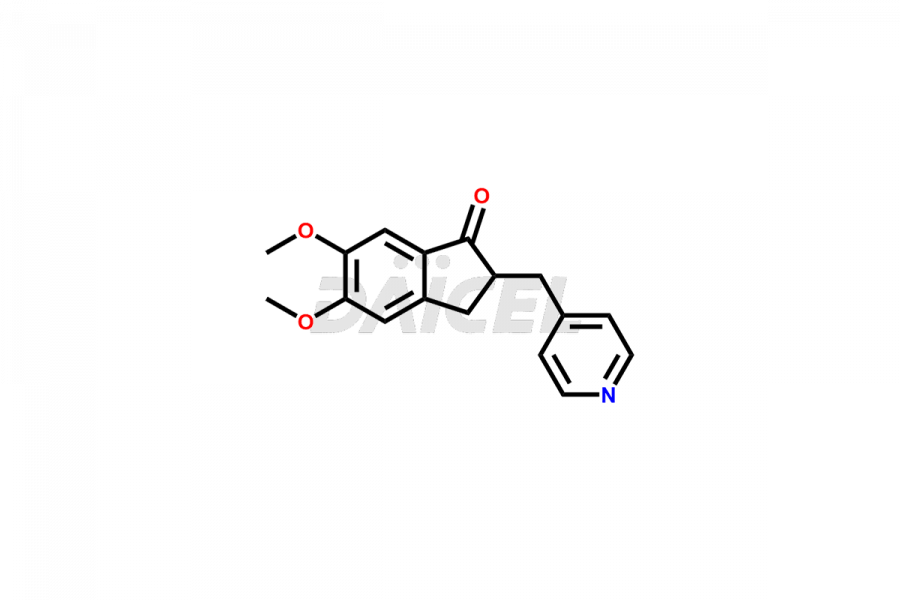
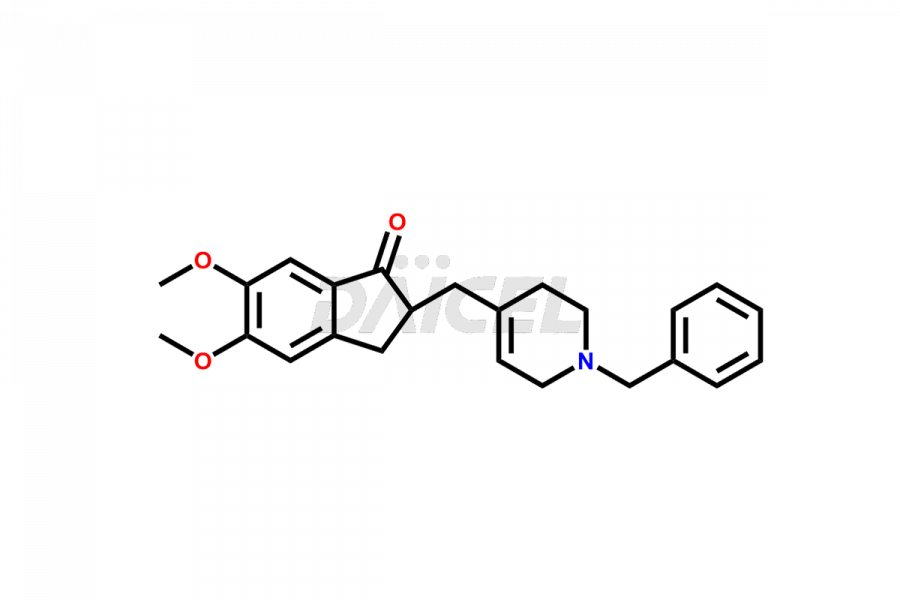
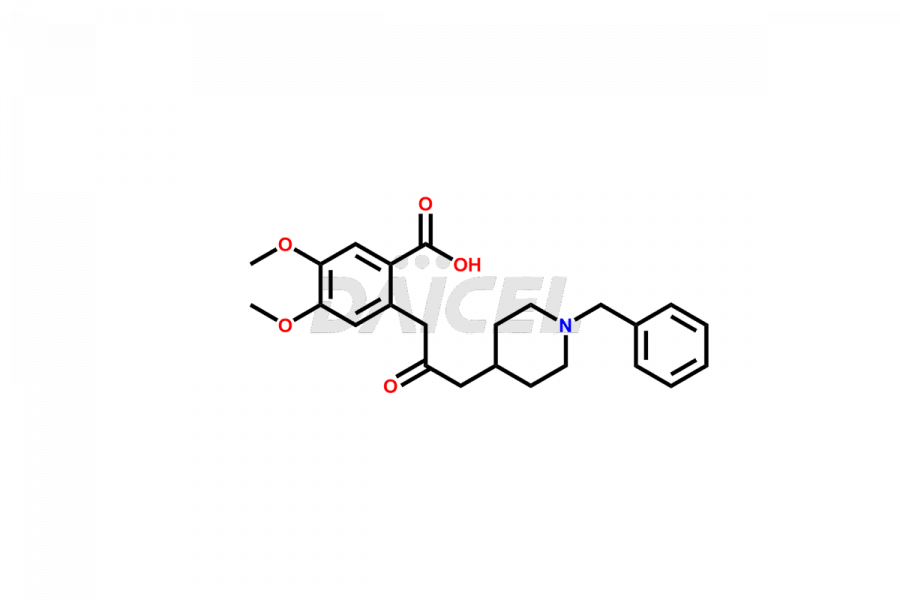
General Information
Donepezil Impurities and Donepezil
Daicel Pharma offers high-quality Donepezil impurities such as Donepezil EP Impurity H and Donepezil Open Ring. They are vital for evaluating the quality, stability, and biological safety of Donepezil. Furthermore, Daicel Pharma specializes in the custom synthesis of Donepezil impurities and ensures their worldwide delivery.
Donepezil [CAS: 120014-06-4] is an oral medicine for Alzheimer’s disease treatment. It is a racemate of equimolar amounts of (R)- and (S)-Donepezil. It reversibly inhibits acetylcholinesterase, primarily aiming to elevate cortical acetylcholine levels in Alzheimer’s therapy.
Donepezil: Use and Commercial Availability
Donepezil, in its hydrochloride form, is a piperidine derivative. It is for treating Alzheimer’s disease. It also treats dementia in mild and moderate forms. Donepezil is marketed under the brand names Aricept and Adlarity.
Donepezil Structure and Mechanism of Action

The chemical name of Donepezil is 2,3-Dihydro-5,6-dimethoxy-2-[[1-(phenylmethyl)-4-piperidinyl]methyl]-1H-inden-1-one. The chemical formula for Donepezil is C24H29NO3, and its molecular weight is approximately 379.5g/mol.
Donepezil is a centrally acting, reversible acetylcholinesterase inhibitor that breaks down acetylcholine after its release from the presynapse. It prevents acetylcholine hydrolysis, thereby increasing the availability of acetylcholine at the synapses. Further, it enhances cholinergic transmission.
Donepezil Impurities and Synthesis
During the synthesis1 of Donepezil, precise measures can uphold its purity and potency. Despite these precautions, impurities can occur, affecting the drug’s quality. Advanced purification techniques such as chromatography and crystallization can mitigate impurities, preserving the integrity of Donepezil.
Daicel Pharma offers a comprehensive Certificate of Analysis (CoA) for Donepezil impurities, such as Donepezil EP Impurity H and Donepezil Open Ring. The CoA is from a cGMP-compliant analytical facility and encompasses complete characterization data such as 1H NMR, 13C NMR, IR, MASS, and HPLC purity2. We give additional data, like 13C-DEPT and CHN, on request. Daicel Pharma can also prepare any unidentified Donepezil impurity or degradation product. Furthermore, Daicel Pharma offers highly purified isotope-labeled standards of Donepezil for bioanalytical research and BA/BE studies. We give a complete characterization report upon delivery.