General Information
Cefazolin Impurities and Cefazolin
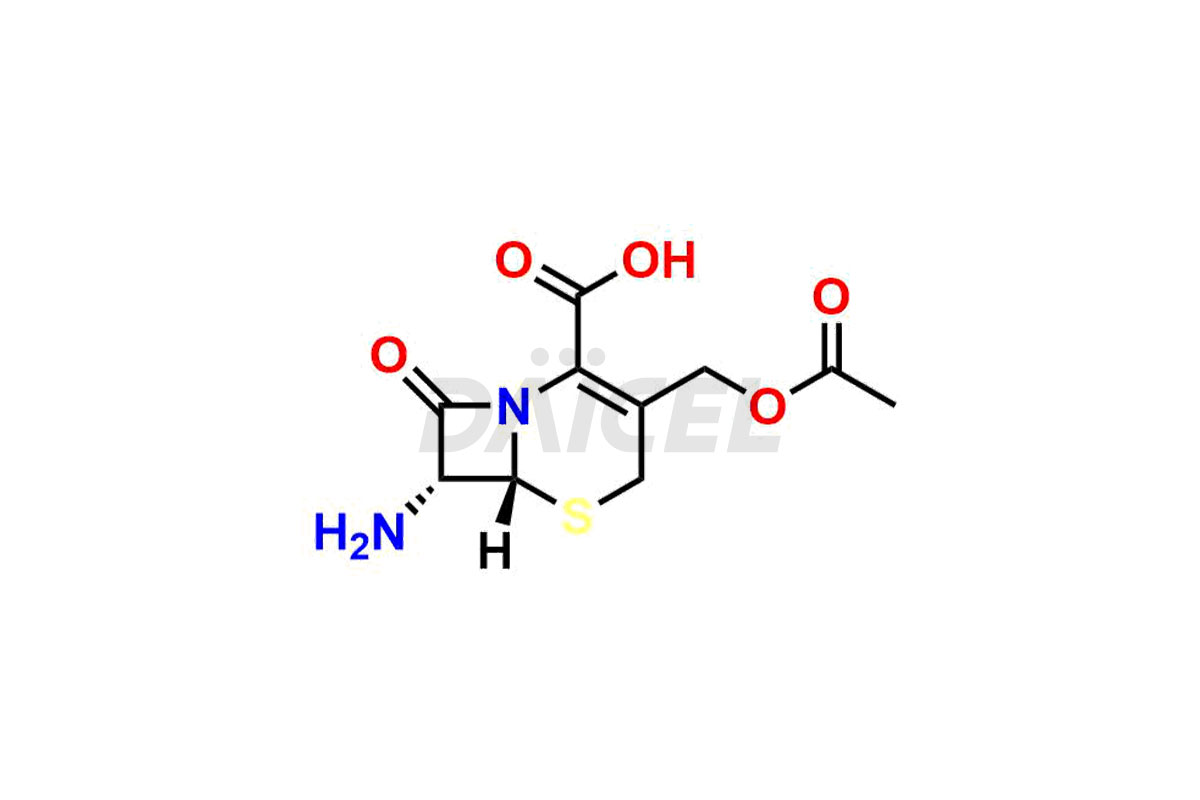
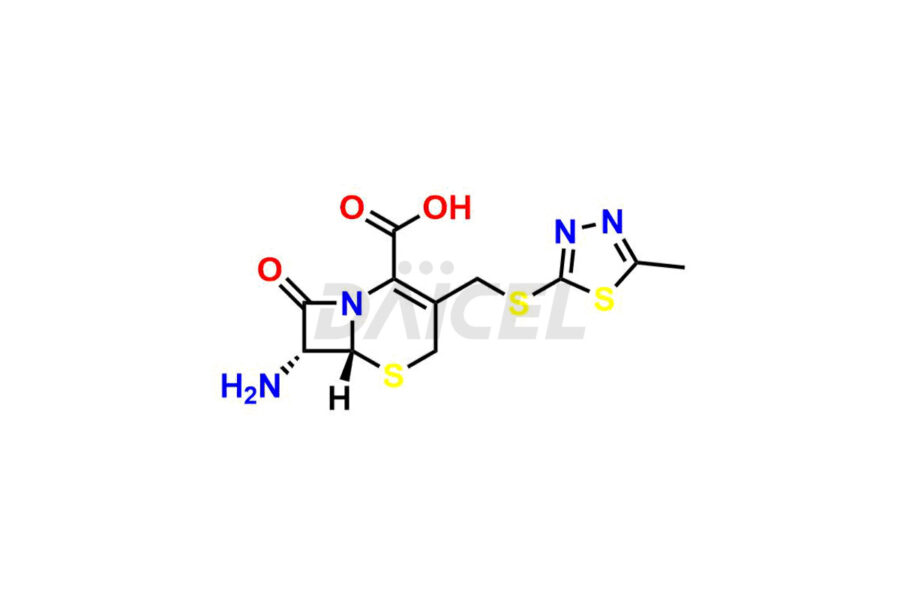
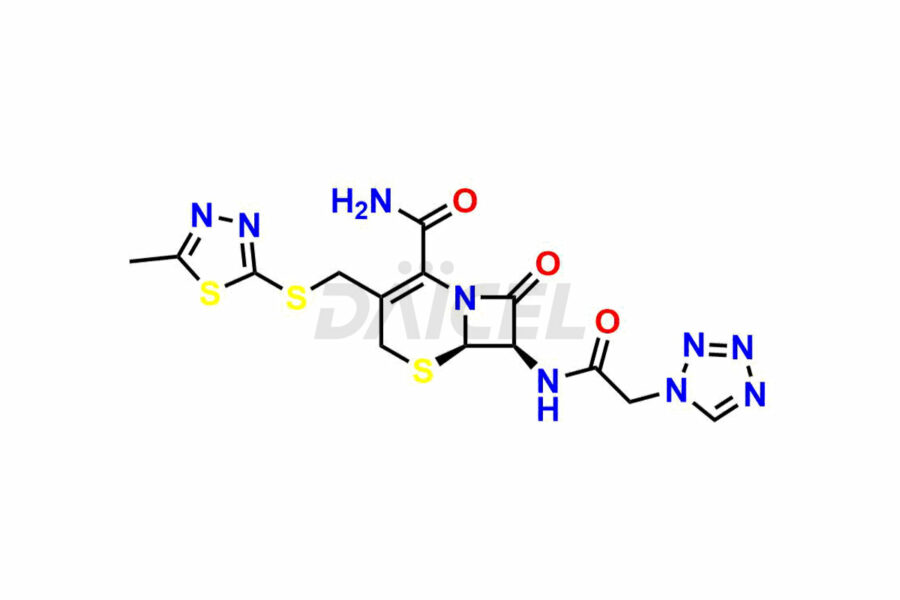
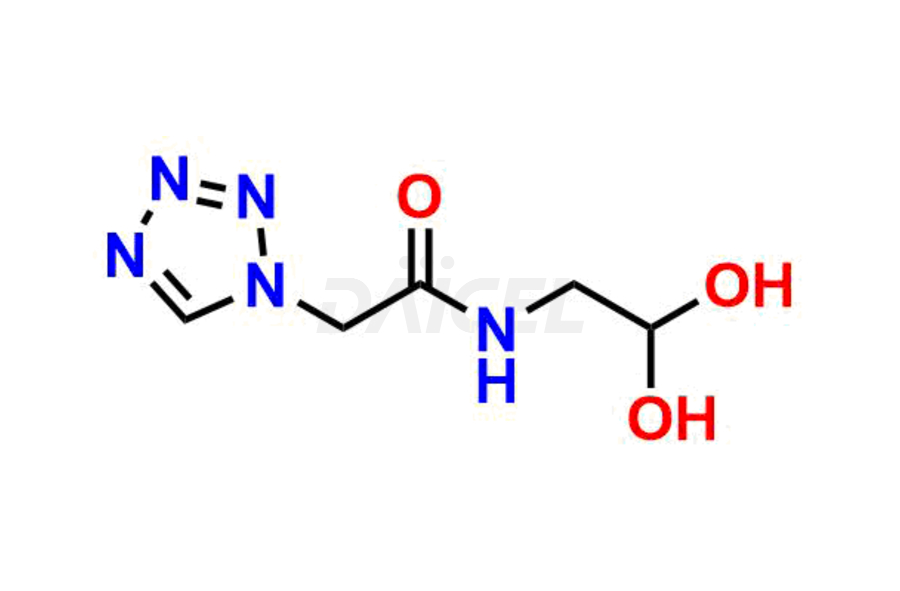
Daicel Pharma synthesizes high-quality Cefazolin impurities, 7-aminocephalosporanic acid, Cefazolin EP impurity A, Cefazolin EP Impurity-K and Tetrazolyl Acetamide Acetal, which are crucial in the analysis of the quality, stability, and biological safety of the active pharmaceutical ingredient, Cefazolin. Moreover, Daicel Pharma offers custom synthesis of Cefazolin impurities and delivers them globally.
Cefazolin [CAS: 25953-19-9] is an antibiotic belonging to beta-lactam and first-generation cephalosporin. It has bactericidal activity.
Cefazolin: Use and Commercial Availability
Cefazolin is an antibiotic primarily for treating bacterial skin infections and moderately severe bacterial infections of the lung, bone, joint, stomach, blood, heart valve, and urinary tract. It is effective against Gram-positive bacteria, specifically staphylococci and streptococci species. The medicine is available under several trade names, including ANCEF, CEFAZOLIN SODIUM, and KEFZOL.
Cefazolin Structure and Mechanism of Action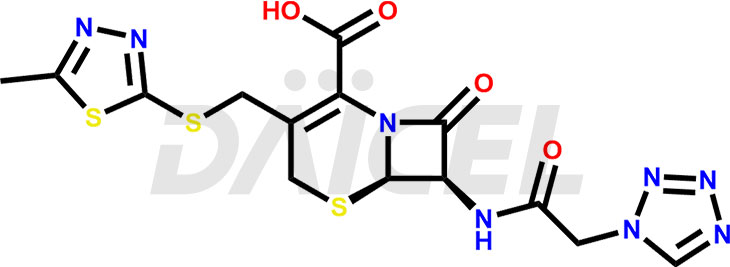
The chemical name of Cefazolin is (6R,7R)-3-[[(5-Methyl-1,3,4-thiadiazol-2-yl)thio]methyl]-8-oxo-7-[[2-(1H-tetrazol-1-yl)acetyl]amino]-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid. Its chemical formula is C14H14N8O4S3, and its molecular weight is approximately 454.5 g/mol.
Cefazolin binds to penicillin-binding proteins (PBP) present on the inner membrane of bacterial cell walls, causing the weakening of the bacterial cell wall and cell lysis.
Cefazolin Impurities and Synthesis
The synthesis1,2 of Cefazolin can lead to the formation of impurities, including epimers, degradation products and more, that can affect the quality and safety of the drug. Manufacturers must employ strict quality control measures to minimize the formation of impurities and conduct regular testing to monitor impurity levels in Cefazolin.
Daicel offers a Certificate of Analysis (CoA) from a cGMP-compliant analytical facility for Cefazolin impurity standards, 7-aminocephalosporanic acid, Cefazolin EP impurity A, Cefazolin EP Impurity-K and Tetrazolyl Acetamide Acetal. The CoA includes complete characterization data, such as 1H NMR, 13C NMR3, IR, MASS, and HPLC purity. We also provide 13C-DEPT and CHN on request. We give a complete characterization report on delivery. Daicel has the technology and expertise to prepare any unknown Cefazolin impurity or degradation product.