General Information
Bilastine Impurities and Bilastine
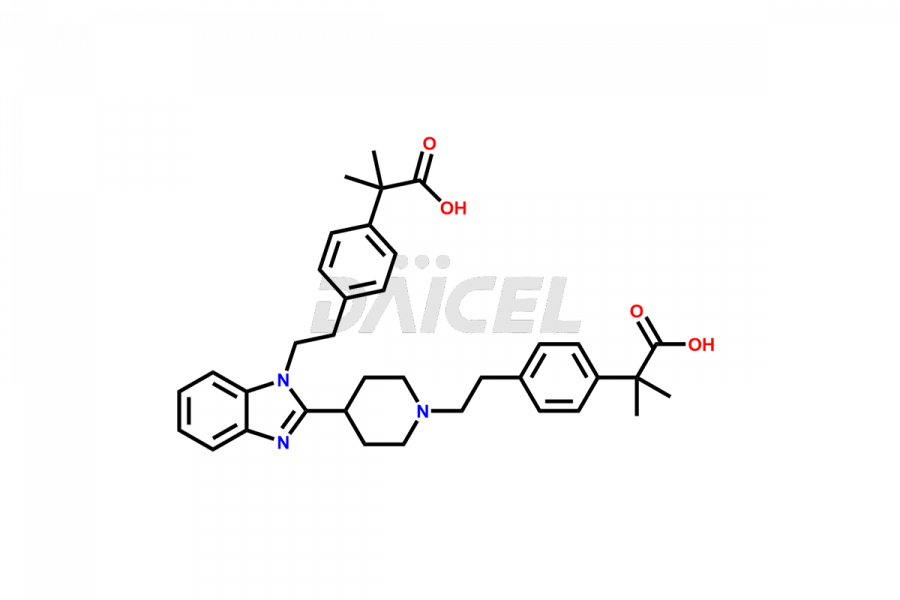
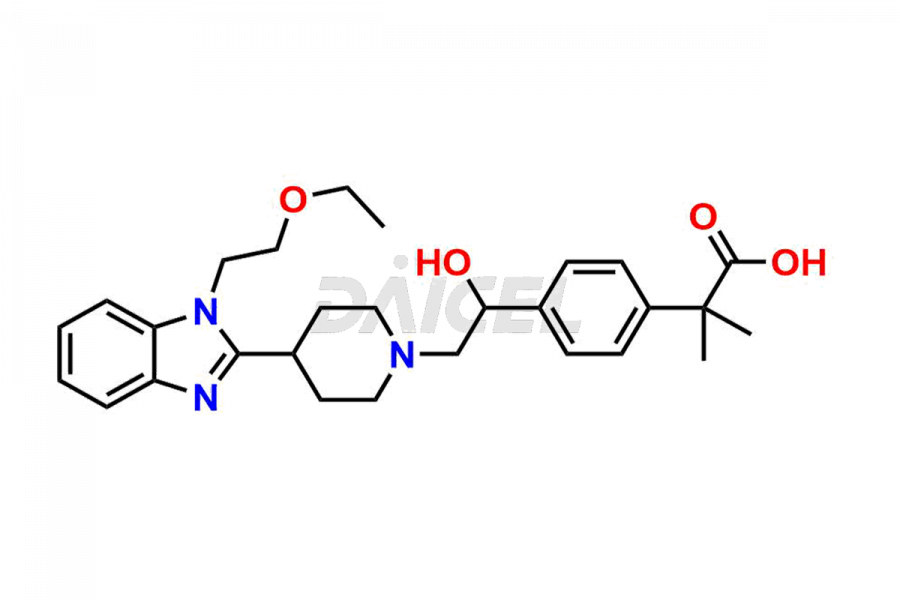
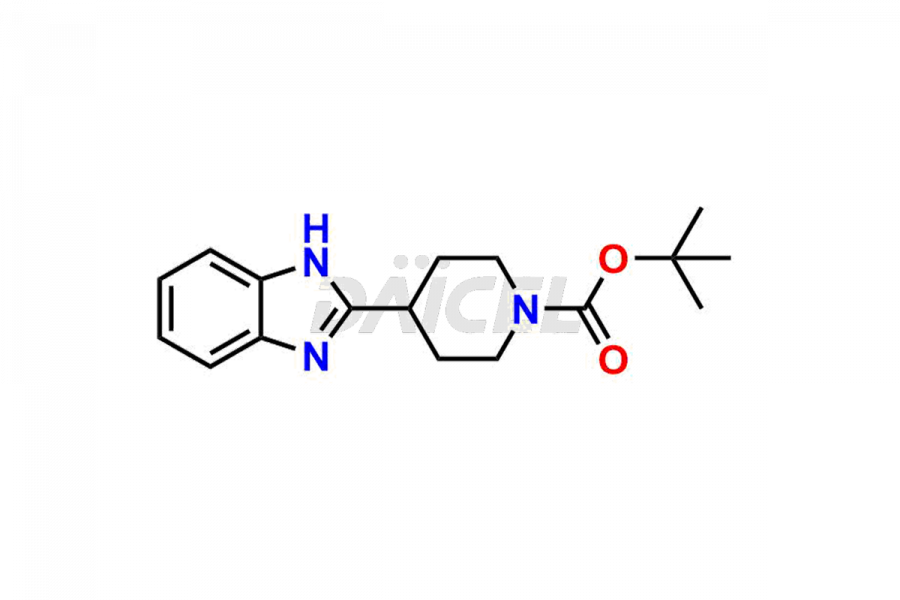
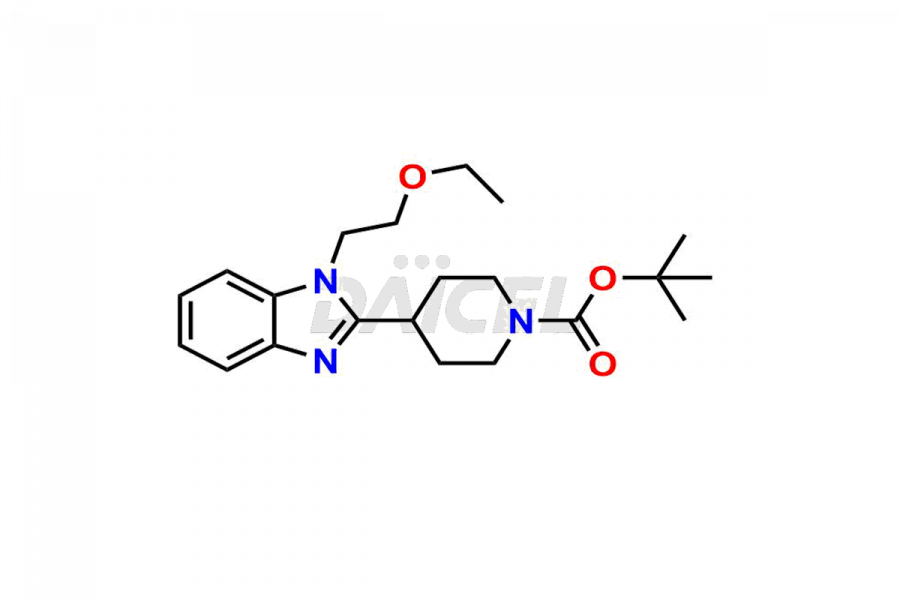
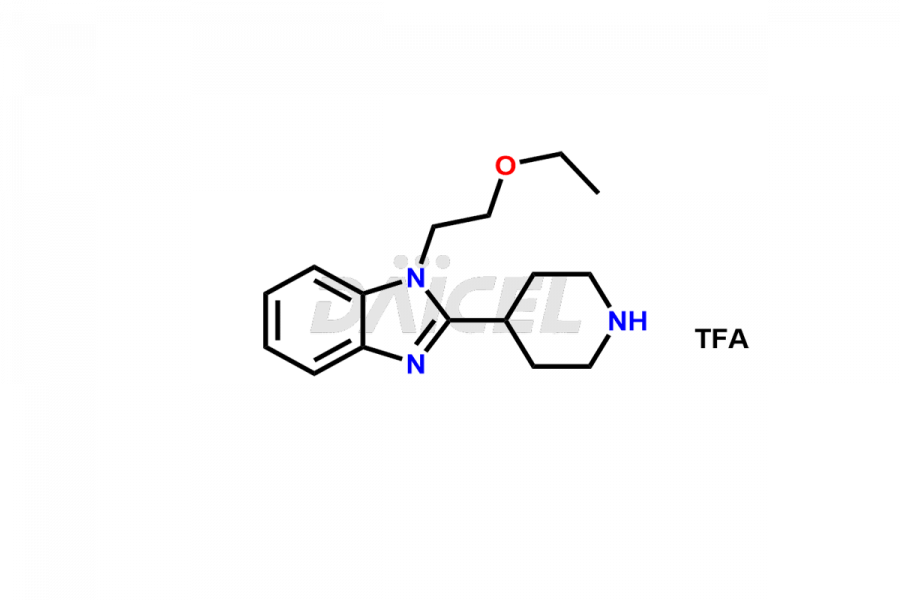
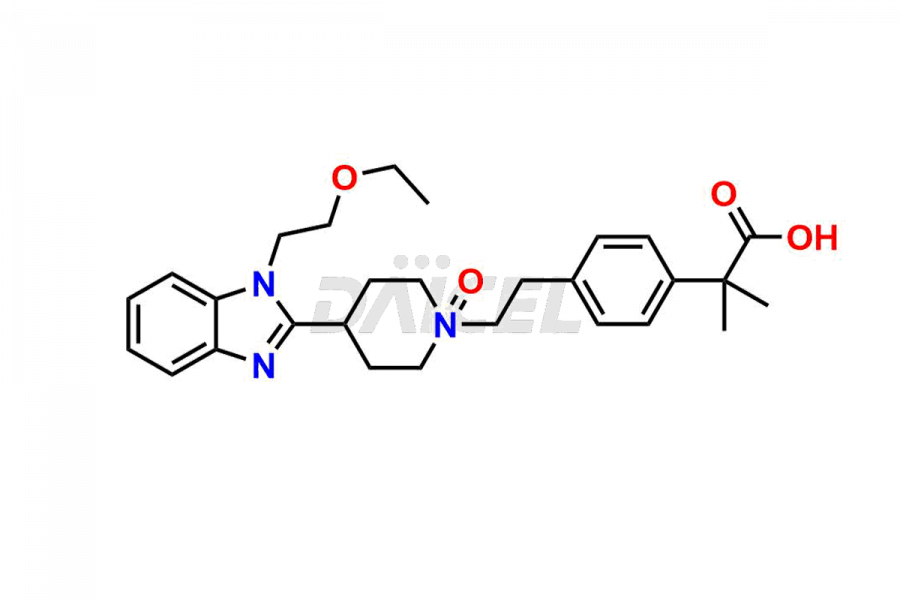
Daicel Pharma synthesizes high-quality Bilastine impurities, including 1′-Hydroxy Bilastine, Bilastine Impurity 1, Bilastine Impurity 2, Bilastine Impurity 7, and Bilastine N-Oxide Impurity. These impurities are essential for evaluating the quality, stability, and safety of Bilastine, an active pharmaceutical ingredient. Additionally, Daicel Pharma offers a customized synthesis of Bilastine impurities for delivery globally to meet the specific needs of our customers.
Bilastine [CAS: 202189-78-4] is an antihistamine that exhibits high selectivity for the H1 histamine receptor antagonist, providing rapid onset of action and long-lasting effects. This medication belongs to the benzimidazole class. Bilastine treats allergic rhinitis and urticaria associated with excessive histamine release.
Bilastine: Use and Commercial Availability
Bilastine is a highly effective, non-sedating, second-generation, H1 receptor antihistamine treating allergic rhinoconjunctivitis and chronic urticaria in patients. Bilastine is available under various brands such as Allertine, Bilargo, Blexten, Bilaxten, Clatra, Ilaxten, etc.
Bilastine Structure and Mechanism of Action 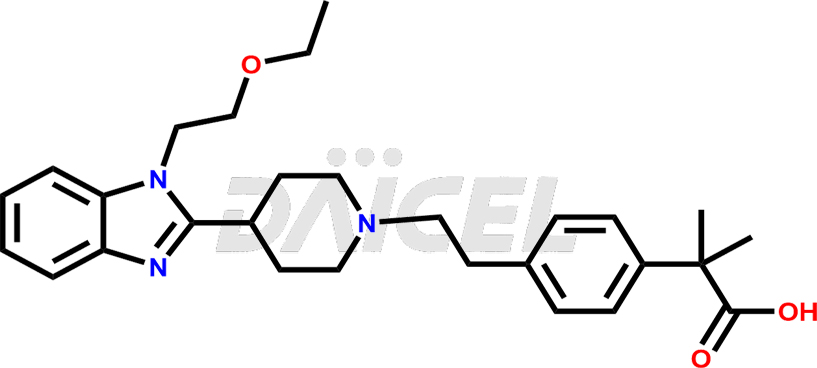
The chemical name of Bilastine is 4-[2-[4-[1-(2-Ethoxyethyl)-1H-benzimidazol-2-yl]-1-piperidinyl]ethyl]-α,α-dimethylbenzeneacetic acid. Its chemical formula is C28H37N3O3, and its molecular weight is approximately 463.6 g/mol.
Bilastine prevents the activation of H1 receptors and releases histamine from mast cells, thus reducing allergic symptoms.
Bilastine Impurities and Synthesis
During the manufacturing1 of Bilastine, impurities may form due to factors such as impure starting materials, reagents, or processing conditions. These impurities can affect the safety and efficacy of the drug. So it is essential to control them within acceptable limits during its manufacturing. Quality control measures, such as analytical testing and process optimization, are necessary to ensure the purity and quality of Bilastine for safe and effective use in patients.
Daicel Pharma provides a Certificate of Analysis (CoA) for Bilastine impurity standards, including 1′-Hydroxy Bilastine, Bilastine Impurity 1, Bilastine Impurity 2, Bilastine Impurity 7, and Bilastine N-Oxide Impurity. The CoA is generated from a cGMP-compliant analytical facility and includes comprehensive characterization data such as 1H NMR, 13C NMR, IR, MASS, and HPLC purity2,3. We can also give additional characterization data like 13C-DEPT and CHN on request. Daicel Pharma is capable of creating unknown Bilastine impurities or degradation products. Each delivery has a complete characterization report.